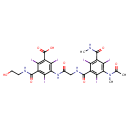| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 03:42:02 UTC |
|---|
| Update Date | 2016-11-09 01:15:27 UTC |
|---|
| Accession Number | CHEM016739 |
|---|
| Identification |
|---|
| Common Name | Ioxaglic acid |
|---|
| Class | Small Molecule |
|---|
| Description | A benzenedicarboxamide compound having N-substituted carbamoyl groups at the 1- and 3-positions, iodo substituents at the 2-, 4- and 6-positions and an acetyl(methyl)amino group at the 5-position. |
|---|
| Contaminant Sources | - STOFF IDENT Compounds
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Acide ioxaglique | ChEBI | | Acido ioxaglico | ChEBI | | Acidum ioxaglicum | ChEBI | | Ioxaglate | ChEBI | | N-(2-Hydroxyethyl)-2,4,6-triiodo-5-(2-(2,4,6-triiodo-3-(N-methylacetamido)-5-(methylcarbamoyl)benzamido)acetamido)isophthalamic acid | ChEBI | | Hexabrix 320 | Kegg | | N-(2-Hydroxyethyl)-2,4,6-triiodo-5-(2-(2,4,6-triiodo-3-(N-methylacetamido)-5-(methylcarbamoyl)benzamido)acetamido)isophthalamate | Generator | | Hexabrix | MeSH | | Ioxaglate meglumine | MeSH | | Ioxaglate sodium | MeSH | | Ioxaglate, methylglucamine | MeSH | | Ioxaglic acid monosodium salt | MeSH | | Ioxaglic acid, calcium salt (2:1) | MeSH | | Meglumine, ioxaglate | MeSH | | Methylglucamine ioxaglate | MeSH | | p-286 (Contrast media) | MeSH | | P286 (Contrast media) | MeSH | | Ioxaglic acid | MeSH |
|
|---|
| Chemical Formula | C24H21I6N5O8 |
|---|
| Average Molecular Mass | 1268.886 g/mol |
|---|
| Monoisotopic Mass | 1268.566 g/mol |
|---|
| CAS Registry Number | 59017-64-0 |
|---|
| IUPAC Name | 3-[(2-hydroxyethyl)carbamoyl]-2,4,6-triiodo-5-(2-{[2,4,6-triiodo-3-(methylcarbamoyl)-5-(N-methylacetamido)phenyl]formamido}acetamido)benzoic acid |
|---|
| Traditional Name | ioxaglate |
|---|
| SMILES | CNC(=O)C1=C(I)C(C(=O)NCC(=O)NC2=C(I)C(C(O)=O)=C(I)C(C(=O)NCCO)=C2I)=C(I)C(N(C)C(C)=O)=C1I |
|---|
| InChI Identifier | InChI=1S/C24H21I6N5O8/c1-7(37)35(3)20-17(29)10(21(39)31-2)13(25)11(18(20)30)23(41)33-6-8(38)34-19-15(27)9(22(40)32-4-5-36)14(26)12(16(19)28)24(42)43/h36H,4-6H2,1-3H3,(H,31,39)(H,32,40)(H,33,41)(H,34,38)(H,42,43) |
|---|
| InChI Key | TYYBFXNZMFNZJT-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as acylaminobenzoic acid and derivatives. These are derivatives of amino benzoic acid derivatives where the amine group is N-acylated. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Benzoic acids and derivatives |
|---|
| Direct Parent | Acylaminobenzoic acid and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Hippuric acid or derivatives
- N-acyl-alpha amino acid or derivatives
- Acylaminobenzoic acid or derivatives
- Alpha-amino acid amide
- O-haloacetanilide
- P-haloacetanilide
- Haloacetanilide
- N-substituted-alpha-amino acid
- 2-halobenzoic acid or derivatives
- 4-halobenzoic acid or derivatives
- Halobenzoic acid or derivatives
- 2-halobenzoic acid
- 4-halobenzoic acid
- Halobenzoic acid
- Acetanilide
- Alpha-amino acid or derivatives
- Anilide
- Benzoic acid
- Benzamide
- N-acylethanolamine
- N-arylamide
- Benzoyl
- 1-carboxy-2-haloaromatic compound
- Halobenzene
- Iodobenzene
- Aryl iodide
- Aryl halide
- Vinylogous halide
- Tertiary carboxylic acid amide
- Acetamide
- Carboxamide group
- Secondary carboxylic acid amide
- Carboxylic acid derivative
- Carboxylic acid
- Alkanolamine
- Monocarboxylic acid or derivatives
- Alcohol
- Carbonyl group
- Organohalogen compound
- Organoiodide
- Organonitrogen compound
- Organooxygen compound
- Primary alcohol
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-qTof , Positive | splash10-014i-2960000000-3f14b9f983cc6e713b6a | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-01t9-2449844111-e90290a3b22dc25a5137 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0wmr-2090008000-f5bb9fb25f28462e0f1f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0w29-3030039000-56447aeb18e7372e8f79 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01q1-3200094000-c7581ed2e5b4c5d11722 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-01ba-1190002000-93e912290c5aa896de48 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0abm-9140003000-6c548525ca3ece24c3d6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0596-9000012000-4a1725a31168f144ef7c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0090000000-7f6dea4df1b08916e81a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ldi-0090002000-60710640eda2065ee8c0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03di-2520049000-3278df6ee9d0656c66a0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-01b9-0090001000-804daea98e75716affa6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-017i-1690000000-cd43b626614eaf0d734c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00or-4730095000-f5ec366f7bc25c3bae98 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB09313 |
|---|
| HMDB ID | HMDB0253554 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Ioxaglic_acid |
|---|
| Chemspider ID | 3611 |
|---|
| ChEBI ID | 31718 |
|---|
| PubChem Compound ID | 3742 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|