| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 03:41:05 UTC |
|---|
| Update Date | 2016-11-09 01:15:27 UTC |
|---|
| Accession Number | CHEM016707 |
|---|
| Identification |
|---|
| Common Name | Felodipine |
|---|
| Class | Small Molecule |
|---|
| Description | Felodipine is a long-acting 1,4-dihydropyridine calcium channel blocker (CCB)b. It acts primarily on vascular smooth muscle cells by stabilizing voltage-gated L-type calcium channels in their inactive conformation. By inhibiting the influx of calcium in smooth muscle cells, felodipine prevents calcium-dependent myocyte contraction and vasoconstriction. Felodipine is the most potent CCB in use and is unique in that it exhibits fluorescent activity. In addition to binding to L-type calcium channels, felodipine binds to a number of calcium-binding proteins, exhibits competitive antagonism of the mineralcorticoid receptor, inhibits the activity of calmodulin-dependent cyclic nucleotide phosphodiesterase, and blocks calcium influx through voltage-gated T-type calcium channels. Felodipine is used to treat mild to moderate essential hypertension. |
|---|
| Contaminant Sources | - HMDB Contaminants - Urine
- STOFF IDENT Compounds
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (+-)-Ethyl methyl 4-(2,3-dichlorophenyl)-1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylate | ChEBI | | 3-Ethyl 5-methyl 4-(2,3-dichlorophenyl)-2,6-dimethyl-1,4-dihydro-3,5-pyridinedicarboxylate | ChEBI | | 4-(2,3-Dichlorophenyl)-1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylic acid ethyl methyl ester | ChEBI | | Felodipina | ChEBI | | Felodipinum | ChEBI | | Plendil | Kegg | | (+-)-Ethyl methyl 4-(2,3-dichlorophenyl)-1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylic acid | Generator | | 3-Ethyl 5-methyl 4-(2,3-dichlorophenyl)-2,6-dimethyl-1,4-dihydro-3,5-pyridinedicarboxylic acid | Generator | | 4-(2,3-Dichlorophenyl)-1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylate ethyl methyl ester | Generator | | DL-Felodipine | HMDB | | 1a Brand OF felodipine | HMDB | | Alpharma brand OF felodipine | HMDB | | AstraZeneca brand OF felodipine | HMDB | | Azupharma brand OF felodipine | HMDB | | Felo biochemie | HMDB | | Felo puren | HMDB | | Felobeta | HMDB | | Felodur | HMDB | | Heumann brand OF felodipine | HMDB | | Hoechst brand OF felodipine | HMDB | | Pharmacia spain brand OF felodipine | HMDB | | TheraPharm brand OF felodipine | HMDB | | Felo-puren | HMDB | | Felocor | HMDB | | Felodipin heumann | HMDB | | Felodipin stada | HMDB | | Felodipin dura | HMDB | | Munobal | HMDB | | Pharmaceutica astra brand OF felodipine | HMDB | | Wörwag brand OF felodipine | HMDB | | CT Arzneimittel brand OF felodipine | HMDB | | CT-Arzneimittel brand OF felodipine | HMDB | | Aliud brand OF felodipine | HMDB | | Alphapharm brand OF felodipine | HMDB | | Astra brand OF felodipine | HMDB | | BC Brand OF felodipine | HMDB | | Felodipin al | HMDB | | Felodipin azu | HMDB | | Felodipin ratiopharm | HMDB | | Merck dura brand OF felodipine | HMDB | | Perfudal | HMDB | | Promed brand OF felodipine | HMDB | | Stadapharm brand OF felodipine | HMDB | | Betapharm brand OF felodipine | HMDB | | Felodipin von CT | HMDB | | AbZ brand OF felodipine | HMDB | | Agon | HMDB | | Aventis brand OF felodipine | HMDB | | Felodipin 1a pharma | HMDB | | Felodipin abz | HMDB | | Felodipin-ratiopharm | HMDB | | Felogamma | HMDB | | Fensel | HMDB | | Flodil | HMDB | | Heumann, felodipin | HMDB | | Hexal brand OF felodipine | HMDB | | Modip | HMDB | | Renedil | HMDB | | Ratiopharm brand OF felodipine | HMDB | | Von CT, felodipin | HMDB |
|
|---|
| Chemical Formula | C18H19Cl2NO4 |
|---|
| Average Molecular Mass | 384.254 g/mol |
|---|
| Monoisotopic Mass | 383.069 g/mol |
|---|
| CAS Registry Number | 72509-76-3 |
|---|
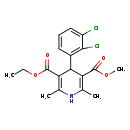
| IUPAC Name | 3-ethyl 5-methyl 4-(2,3-dichlorophenyl)-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate |
|---|
| Traditional Name | felodipine |
|---|
| SMILES | CCOC(=O)C1=C(C)NC(C)=C(C1C1=C(Cl)C(Cl)=CC=C1)C(=O)OC |
|---|
| InChI Identifier | InChI=1S/C18H19Cl2NO4/c1-5-25-18(23)14-10(3)21-9(2)13(17(22)24-4)15(14)11-7-6-8-12(19)16(11)20/h6-8,15,21H,5H2,1-4H3 |
|---|
| InChI Key | RZTAMFZIAATZDJ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as dihydropyridinecarboxylic acids and derivatives. Dihydropyridinecarboxylic acids and derivatives are compounds containing a dihydropyridine moiety bearing a carboxylic acid group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Pyridines and derivatives |
|---|
| Sub Class | Hydropyridines |
|---|
| Direct Parent | Dihydropyridinecarboxylic acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Dihydropyridinecarboxylic acid derivative
- 1,2-dichlorobenzene
- Chlorobenzene
- Halobenzene
- Aryl chloride
- Aryl halide
- Monocyclic benzene moiety
- Dicarboxylic acid or derivatives
- Benzenoid
- Vinylogous amide
- Alpha,beta-unsaturated carboxylic ester
- Enoate ester
- Methyl ester
- Amino acid or derivatives
- Carboxylic acid ester
- Carboxylic acid derivative
- Secondary aliphatic amine
- Enamine
- Secondary amine
- Azacycle
- Organonitrogen compound
- Hydrocarbon derivative
- Organooxygen compound
- Organic oxide
- Amine
- Organohalogen compound
- Carbonyl group
- Organopnictogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Organochloride
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-01t9-3009000000-5b611cdc2d075de69610 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-qTof , Positive | splash10-000j-0922000000-9b26b987caa87b8a13af | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-0udu-0139000000-085071c20c5d0c134d8f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001r-0019000000-33bf0db3bf36f29993fb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0fe0-0095000000-c27a529dc8d62e0c3663 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004i-0490000000-b027fd5c39a74d5a8983 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0009000000-74fea585f5eab97e301d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01q9-0029000000-611d97581685b1d82833 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0wbc-1095000000-178af93061f6d2ad0db1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0019-0009000000-88f48d1738d7c1f69a11 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0bu9-0129000000-d31431a7eda7ca7396bc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0piu-0982000000-98f7be8fe67f962d9ef1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0009000000-e514f5ea1713c07d88b1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001i-2009000000-409c00755f945e97a932 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-001i-9280000000-97534f29e936d15877c7 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB01023 |
|---|
| HMDB ID | HMDB0015158 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Felodipine |
|---|
| Chemspider ID | 3216 |
|---|
| ChEBI ID | 585948 |
|---|
| PubChem Compound ID | 3333 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|