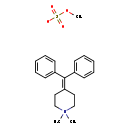| 4-(Diphenylmethylene)-1,1-dimethylpiperidinium methyl sulfate | ChEBI |
| Diphemanil methosulfate | ChEBI |
| Diphemanil metilsulfate | ChEBI |
| Diphemanili metilsulfas | ChEBI |
| Metilsulfate de diphemanil | ChEBI |
| Metilsulfato de difemanilo | ChEBI |
| N,N-Dimethyl-4-piperidylidene-1,1-diphenylmethane methylsulfate | ChEBI |
| p-(alpha-Phenylbenzylidene)-1,1-dimethylpiperidinium methyl sulfate | ChEBI |
| Prantal | Kegg |
| 4-(Diphenylmethylene)-1,1-dimethylpiperidinium methyl sulfuric acid | Generator |
| 4-(Diphenylmethylene)-1,1-dimethylpiperidinium methyl sulphate | Generator |
| 4-(Diphenylmethylene)-1,1-dimethylpiperidinium methyl sulphuric acid | Generator |
| Diphemanil methosulfuric acid | Generator |
| Diphemanil methosulphate | Generator |
| Diphemanil methosulphuric acid | Generator |
| Diphemanil metilsulfuric acid | Generator |
| Diphemanil metilsulphate | Generator |
| Diphemanil metilsulphuric acid | Generator |
| Diphemanili metilsulphas | Generator |
| Metilsulfuric acid de diphemanil | Generator |
| Metilsulphate de diphemanil | Generator |
| Metilsulphuric acid de diphemanil | Generator |
| Metilsulphato de difemanilo | Generator |
| N,N-Dimethyl-4-piperidylidene-1,1-diphenylmethane methylsulfuric acid | Generator |
| N,N-Dimethyl-4-piperidylidene-1,1-diphenylmethane methylsulphate | Generator |
| N,N-Dimethyl-4-piperidylidene-1,1-diphenylmethane methylsulphuric acid | Generator |
| p-(a-Phenylbenzylidene)-1,1-dimethylpiperidinium methyl sulfate | Generator |
| p-(a-Phenylbenzylidene)-1,1-dimethylpiperidinium methyl sulfuric acid | Generator |
| p-(a-Phenylbenzylidene)-1,1-dimethylpiperidinium methyl sulphate | Generator |
| p-(a-Phenylbenzylidene)-1,1-dimethylpiperidinium methyl sulphuric acid | Generator |
| p-(alpha-Phenylbenzylidene)-1,1-dimethylpiperidinium methyl sulfuric acid | Generator |
| p-(alpha-Phenylbenzylidene)-1,1-dimethylpiperidinium methyl sulphate | Generator |
| p-(alpha-Phenylbenzylidene)-1,1-dimethylpiperidinium methyl sulphuric acid | Generator |
| p-(Α-phenylbenzylidene)-1,1-dimethylpiperidinium methyl sulfate | Generator |
| p-(Α-phenylbenzylidene)-1,1-dimethylpiperidinium methyl sulfuric acid | Generator |
| p-(Α-phenylbenzylidene)-1,1-dimethylpiperidinium methyl sulphate | Generator |
| p-(Α-phenylbenzylidene)-1,1-dimethylpiperidinium methyl sulphuric acid | Generator |
| Diphemanil methylsulfuric acid | Generator |
| Diphemanil methylsulphate | Generator |
| Diphemanil methylsulphuric acid | Generator |