| Synonyms | | Value | Source |
|---|
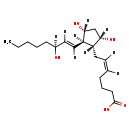
| (+)-Prostaglandin F2a | ChEBI | | (5Z,13E)-(15S)-9alpha,11alpha,15-Trihydroxyprosta-5,13-dienoate | ChEBI | | 7-[3,5-Dihydroxy-2-(3-hydroxy-1-octenyl)cyclopentyl]-5-heptenoic acid | ChEBI | | 9,11,15-Trihydroxy-(5Z,9a,11a,13E,15S)-prosta-5,13-dien-1-Oic acid | ChEBI | | 9a,11a-PGF2 | ChEBI | | Amoglandin | ChEBI | | Cyclosin | ChEBI | | Dinoprosta | ChEBI | | Dinoprostum | ChEBI | | Enzaprost | ChEBI | | Enzaprost F | ChEBI | | L-PGF2-alpha | ChEBI | | L-Prostaglandin F2-alpha | ChEBI | | Panacelan | ChEBI | | PGF2a | ChEBI | | PGF2alpha | ChEBI | | Prostaglandin F2a | ChEBI | | Prostin F 2 alpha | ChEBI | | Protamodin | ChEBI | | U 14583 | ChEBI | | (5Z,9alpha,11alpha,13E,15S)-9,11,15-Trihydroxyprosta-5,13-dienoate | Kegg | | Prosmon | Kegg | | (5Z,13E)-(15S)-9a,11a,15-Trihydroxyprosta-5,13-dienoate | Generator | | (5Z,13E)-(15S)-9a,11a,15-Trihydroxyprosta-5,13-dienoic acid | Generator | | (5Z,13E)-(15S)-9alpha,11alpha,15-Trihydroxyprosta-5,13-dienoic acid | Generator | | (5Z,13E)-(15S)-9Α,11α,15-trihydroxyprosta-5,13-dienoate | Generator | | (5Z,13E)-(15S)-9Α,11α,15-trihydroxyprosta-5,13-dienoic acid | Generator | | 7-[3,5-Dihydroxy-2-(3-hydroxy-1-octenyl)cyclopentyl]-5-heptenoate | Generator | | 9,11,15-Trihydroxy-(5Z,9a,11a,13E,15S)-prosta-5,13-dien-1-Oate | Generator | | L-PGF2-a | Generator | | L-PGF2-Α | Generator | | L-Prostaglandin F2-a | Generator | | L-Prostaglandin F2-α | Generator | | PGF2Α | Generator | | Prostin F 2 a | Generator | | Prostin F 2 α | Generator | | (5Z,9a,11a,13E,15S)-9,11,15-Trihydroxyprosta-5,13-dienoate | Generator | | (5Z,9a,11a,13E,15S)-9,11,15-Trihydroxyprosta-5,13-dienoic acid | Generator | | (5Z,9alpha,11alpha,13E,15S)-9,11,15-Trihydroxyprosta-5,13-dienoic acid | Generator | | (5Z,9Α,11α,13E,15S)-9,11,15-trihydroxyprosta-5,13-dienoate | Generator | | (5Z,9Α,11α,13E,15S)-9,11,15-trihydroxyprosta-5,13-dienoic acid | Generator | | Dinoprost | ChEBI | | (Z)-7-[(1R,2R,3R,5S)-3,5-Dihydroxy-2-[(e,3S)-3-hydroxyoct-1-enyl]cyclopentyl]hept-5-enoate | Generator | | 9alpha,11beta-PGF2 | MeSH | | Prostaglandin F2 | MeSH | | 9alpha,11beta PGF2 | MeSH | | F2 alpha, Prostaglandin | MeSH | | F2alpha, Prostaglandin | MeSH | | Prostaglandin F2 alpha | MeSH | | Prostaglandin F2alpha | MeSH | | alpha, PGF2 | MeSH | | Estrofan | MeSH | | PGF2 | MeSH | | PGF2 alpha | MeSH |
|
|---|
| InChI Identifier | InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h4,7,12-13,15-19,21-23H,2-3,5-6,8-11,14H2,1H3,(H,24,25)/b7-4-,13-12+/t15-,16+,17+,18-,19+/m0/s1 |
|---|