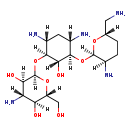
Identification Common Name Dibekacin Class Small Molecule Description A kanamycin that is kanamycin B lacking the 3- and 4-hydroxy groups on the 2,6-diaminosugar ring. Contaminant Sources ToxCast & Tox21 Chemicals Contaminant Type Not Available Chemical Structure Synonyms Value Source 3',4'-Dideoxykanamycin b ChEBI DKM ChEBI O-3-Amino-3-deoxy-alpha-D-glucopyranosyl-(1-4)-O-(2,6-diamino-2,3,4,6-tetradeoxy-alpha-D-erythro-hexopyranosyl-(1-6))-2-deoxy-L-streptamine ChEBI Panamicin ChEBI O-3-Amino-3-deoxy-a-D-glucopyranosyl-(1-4)-O-(2,6-diamino-2,3,4,6-tetradeoxy-a-D-erythro-hexopyranosyl-(1-6))-2-deoxy-L-streptamine Generator O-3-Amino-3-deoxy-α-D-glucopyranosyl-(1-4)-O-(2,6-diamino-2,3,4,6-tetradeoxy-α-D-erythro-hexopyranosyl-(1-6))-2-deoxy-L-streptamine Generator Dibekacin MeSH Dibekacin sulfate MeSH Sulfate, dibekacin MeSH b, 3',4'-Dideoxykanamycin MeSH Dideoxykanamycin b MeSH Orbicin MeSH 3',4' Dideoxykanamycin b MeSH Dideoxykanamycine MeSH Sulphate, dibekacin MeSH Dibekacin sulphate MeSH
Chemical Formula C18 H37 N5 O8 Average Molecular Mass 451.521 g/mol Monoisotopic Mass 451.264 g/mol CAS Registry Number 34493-98-6 IUPAC Name (2S,3R,4S,5S,6R)-4-amino-2-{[(1S,2S,3R,4S,6R)-4,6-diamino-3-{[(2R,3R,6S)-3-amino-6-(aminomethyl)oxan-2-yl]oxy}-2-hydroxycyclohexyl]oxy}-6-(hydroxymethyl)oxane-3,5-diol Traditional Name dibekacin SMILES [H][C@@]1(N)CC[C@@]([H])(CN)O[C@]1([H])O[C@]1([H])[C@@]([H])(N)C[C@@]([H])(N)[C@]([H])(O[C@@]2([H])O[C@]([H])(CO)[C@@]([H])(O)[C@]([H])(N)[C@@]2([H])O)[C@@]1([H])O InChI Identifier InChI=1S/C18H37N5O8/c19-4-6-1-2-7(20)17(28-6)30-15-8(21)3-9(22)16(14(15)27)31-18-13(26)11(23)12(25)10(5-24)29-18/h6-18,24-27H,1-5,19-23H2/t6-,7+,8-,9+,10+,11-,12+,13+,14-,15+,16-,17+,18+/m0/s1 InChI Key JJCQSGDBDPYCEO-XVZSLQNASA-N