| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 03:39:39 UTC |
|---|
| Update Date | 2026-03-26 21:38:31 UTC |
|---|
| Accession Number | CHEM016659 |
|---|
| Identification |
|---|
| Common Name | Desogestrel |
|---|
| Class | Small Molecule |
|---|
| Description | |
|---|
| Contaminant Sources | - HMDB Contaminants - Urine
- STOFF IDENT Compounds
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
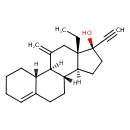
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 13-Ethyl-11-methylene-18,19-dinor-17alpha-pregn-4-en-20-yn-17-ol | ChEBI | | Cerazette | ChEBI | | Desogestrelum | ChEBI | | 13-Ethyl-11-methylene-18,19-dinor-17a-pregn-4-en-20-yn-17-ol | Generator | | 13-Ethyl-11-methylene-18,19-dinor-17α-pregn-4-en-20-yn-17-ol | Generator | | 13 Ethyl 11 methylene 18,19 dinor 17 alpha pregn 4 en 20 yn 17 ol | HMDB | | 13-Ethyl-11-methylene-18,19-dinor-17 alpha-pregn-4-en-20-yn-17-ol | HMDB | | Marvelon | HMDB | | Organon brand OF desogestrel | HMDB | | alpha-Pregn-4-en-20-yn-17-ol, 13-ethyl-11-methylene-18,19-dinor-17 | HMDB |
|
|---|
| Chemical Formula | C22H30O |
|---|
| Average Molecular Mass | 310.473 g/mol |
|---|
| Monoisotopic Mass | 310.230 g/mol |
|---|
| CAS Registry Number | 54024-22-5 |
|---|
| IUPAC Name | (1S,2R,10S,11S,14R,15S)-15-ethyl-14-ethynyl-17-methylidenetetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadec-6-en-14-ol |
|---|
| Traditional Name | mircette |
|---|
| SMILES | [H][C@@]12CC[C@@](O)(C#C)[C@@]1(CC)CC(=C)[C@]1([H])[C@@]3([H])CCCC=C3CC[C@@]21[H] |
|---|
| InChI Identifier | InChI=1S/C22H30O/c1-4-21-14-15(3)20-17-9-7-6-8-16(17)10-11-18(20)19(21)12-13-22(21,23)5-2/h2,8,17-20,23H,3-4,6-7,9-14H2,1H3/t17-,18-,19-,20+,21-,22-/m0/s1 |
|---|
| InChI Key | RPLCPCMSCLEKRS-BPIQYHPVSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as estrane steroids. These are steroids with a structure based on the estrane skeleton. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Estrane steroids |
|---|
| Direct Parent | Estrane steroids |
|---|
| Alternative Parents | |
|---|
| Substituents | - 17-hydroxysteroid
- Hydroxysteroid
- Estrane-skeleton
- Delta-4-steroid
- Ynone
- Tertiary alcohol
- Cyclic alcohol
- Acetylide
- Organic oxygen compound
- Hydrocarbon derivative
- Organooxygen compound
- Alcohol
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | - terminal acetylenic compound (CHEBI:4453 )
- 17beta-hydroxy steroid (CHEBI:4453 )
- C21 steroids (gluco/mineralocorticoids, progestogens) and derivatives (C07629 )
- C21 steroids (gluco/mineralocorticoids, progestogins) and derivatives (LMST02030104 )
|
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-001i-1290000000-9475ca0bf2f8bacf92e6 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-0uxr-2259000000-f9baa1c23b9a2a4c85d3 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0059000000-aa79c27f1567e9a1bbb8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01q9-1191000000-6107987c3071dc2f2bce | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f6x-6490000000-517254a3119708155490 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0009000000-a4421231af9da549d34e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-0049000000-5671ecc974cfc97a058d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-055f-0090000000-f5a84a6463500aef18be | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0009000000-bd627e7955e8c8fb1ae0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-0009000000-bd627e7955e8c8fb1ae0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0bt9-0091000000-dde68f19d9643d308450 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0239000000-4888d713f81fa1d000c4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0159-0951000000-5d7499db9a7f85012ce3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-003r-1930000000-2bccd3c0a6581ed2a815 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB00304 |
|---|
| HMDB ID | HMDB0014449 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Desogestrel |
|---|
| Chemspider ID | 37400 |
|---|
| ChEBI ID | 4453 |
|---|
| PubChem Compound ID | 40973 |
|---|
| Kegg Compound ID | C07629 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. Gentile DM, Verhoeven CH, Shimada T, Back DJ: The role of CYP2C in the in vitro bioactivation of the contraceptive steroid desogestrel. J Pharmacol Exp Ther. 1998 Dec;287(3):975-82. | | 2. Korhonen T, Tolonen A, Uusitalo J, Lundgren S, Jalonen J, Laine K: The role of CYP2C and CYP3A in the disposition of 3-keto-desogestrel after administration of desogestrel. Br J Clin Pharmacol. 2005 Jul;60(1):69-75. | | 3. Simons K, Toomre D: Lipid rafts and signal transduction. Nat Rev Mol Cell Biol. 2000 Oct;1(1):31-9. | | 4. Watson AD: Thematic review series: systems biology approaches to metabolic and cardiovascular disorders. Lipidomics: a global approach to lipid analysis in biological systems. J Lipid Res. 2006 Oct;47(10):2101-11. Epub 2006 Aug 10. | | 5. Sethi JK, Vidal-Puig AJ: Thematic review series: adipocyte biology. Adipose tissue function and plasticity orchestrate nutritional adaptation. J Lipid Res. 2007 Jun;48(6):1253-62. Epub 2007 Mar 20. | | 6. Lingwood D, Simons K: Lipid rafts as a membrane-organizing principle. Science. 2010 Jan 1;327(5961):46-50. doi: 10.1126/science.1174621. | | 7. The lipid handbook with CD-ROM |
|
|---|