| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 03:39:35 UTC |
|---|
| Update Date | 2016-11-09 01:15:26 UTC |
|---|
| Accession Number | CHEM016656 |
|---|
| Identification |
|---|
| Common Name | Delavirdine |
|---|
| Class | Small Molecule |
|---|
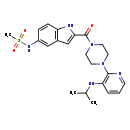
| Description | The amide resulting from the formal condensation of 5--1H-indole-2-carboxylic acid and 4-amino group of 1-piperazine, delavirdine is a non-nucleoside reverse transcriptase inhibitor with activity specific for HIV-1. Viral resistance emerges rapidly when delavirdine is used alone, so it is therefore used (as the methanesulfonic acid salt) with other antiretrovirals for combination therapy of HIV infection. |
|---|
| Contaminant Sources | - HMDB Contaminants - Urine
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (N-[2-[4-[3-(1-Methylethylamino)pyridin-2-yl]piperazin-1-yl]carbonyl-1H-indol-5-yl] methanesulfonamide) | ChEBI | | 1-(3-((1-Methylethyl)amino)-2-pyridinyl)-4-((5-((methylsulfonyl)amino)-1H-indol-2-yl)carbonyl)piperazine | ChEBI | | 2-(4-(5-Methanesulfonamido-1H-indol-2-ylcarbonyl)-1-piperazinyl)-N-(1-methylethyl)-3-pyridinamine | ChEBI | | N-(2-(1-(3-(Isopropylamino)pyridin-2-yl)piperazine-4-carbonyl)-1H-indol-5-yl)methanesulfonamide | ChEBI | | N-{2-[4-(3-isopropylamino-pyridin-2-yl)-piperazine-1-carbonyl]-1H-indol-5-yl}-methanesulfonamide | ChEBI | | (N-[2-[4-[3-(1-Methylethylamino)pyridin-2-yl]piperazin-1-yl]carbonyl-1H-indol-5-yl] methanesulphonamide) | Generator | | 1-(3-((1-Methylethyl)amino)-2-pyridinyl)-4-((5-((methylsulphonyl)amino)-1H-indol-2-yl)carbonyl)piperazine | Generator | | 2-(4-(5-Methanesulphonamido-1H-indol-2-ylcarbonyl)-1-piperazinyl)-N-(1-methylethyl)-3-pyridinamine | Generator | | N-(2-(1-(3-(Isopropylamino)pyridin-2-yl)piperazine-4-carbonyl)-1H-indol-5-yl)methanesulphonamide | Generator | | N-{2-[4-(3-isopropylamino-pyridin-2-yl)-piperazine-1-carbonyl]-1H-indol-5-yl}-methanesulphonamide | Generator | | Mesylate, delavirdine | HMDB | | Pfizer brand OF delavirdine mesilate | HMDB | | Delavirdine mesylate | HMDB | | Rescriptor | HMDB |
|
|---|
| Chemical Formula | C22H28N6O3S |
|---|
| Average Molecular Mass | 456.561 g/mol |
|---|
| Monoisotopic Mass | 456.194 g/mol |
|---|
| CAS Registry Number | 136817-59-9 |
|---|
| IUPAC Name | N-[2-(4-{3-[(propan-2-yl)amino]pyridin-2-yl}piperazine-1-carbonyl)-1H-indol-5-yl]methanesulfonamide |
|---|
| Traditional Name | delavirdine |
|---|
| SMILES | CC(C)NC1=C(N=CC=C1)N1CCN(CC1)C(=O)C1=CC2=C(N1)C=CC(NS(C)(=O)=O)=C2 |
|---|
| InChI Identifier | InChI=1S/C22H28N6O3S/c1-15(2)24-19-5-4-8-23-21(19)27-9-11-28(12-10-27)22(29)20-14-16-13-17(26-32(3,30)31)6-7-18(16)25-20/h4-8,13-15,24-26H,9-12H2,1-3H3 |
|---|
| InChI Key | WHBIGIKBNXZKFE-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenylalanine and derivatives. Phenylalanine and derivatives are compounds containing phenylalanine or a derivative thereof resulting from reaction of phenylalanine at the amino group or the carboxy group, or from the replacement of any hydrogen of glycine by a heteroatom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Phenylalanine and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenylalanine or derivatives
- N-acyl-alpha amino acid or derivatives
- N-acyl-alpha-amino acid
- 3-phenylpropanoic-acid
- Amphetamine or derivatives
- Aromatic monoterpenoid
- P-menthane monoterpenoid
- Monocyclic monoterpenoid
- Monoterpenoid
- Monocyclic benzene moiety
- Benzenoid
- Carboxamide group
- Secondary carboxylic acid amide
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Organic nitrogen compound
- Organonitrogen compound
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Carbonyl group
- Organopnictogen compound
- Organic oxygen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-004i-1869800000-919b00dc1b3b74381f73 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0bt9-1149800000-a0f76f90872cbd99d53c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-1219000000-cb09ea0c014410a3499e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-001r-2920000000-12fdf4d7579108e38ed2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a6r-7011900000-47425ff75ae0b4e2aab5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-9121100000-a64588514bc1394a9cf5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-9200000000-116fecaf8ef4fa6935bc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0000900000-75f8f7bd14bf867bf702 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0010900000-5d8c991ffb8be5810226 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0079-1933300000-5ce651b88994073e97ed | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0010900000-6d8ef33136c03f3466f9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-06r2-0008900000-4a049c134472e688dda8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-9652200000-831ce982d916abfa0628 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB00705 |
|---|
| HMDB ID | HMDB0014843 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | SPP |
|---|
| Wikipedia Link | Delavirdine |
|---|
| Chemspider ID | 5423 |
|---|
| ChEBI ID | 119573 |
|---|
| PubChem Compound ID | 5625 |
|---|
| Kegg Compound ID | C06941 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|