| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 03:39:17 UTC |
|---|
| Update Date | 2016-11-09 01:15:26 UTC |
|---|
| Accession Number | CHEM016645 |
|---|
| Identification |
|---|
| Common Name | Cyclacillin |
|---|
| Class | Small Molecule |
|---|
| Description | A cyclohexylamido analog of penicillanic acid. |
|---|
| Contaminant Sources | - HMDB Contaminants - Urine
- STOFF IDENT Compounds
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
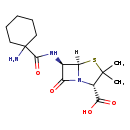
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (1-Aminocyclohexyl)penicillin | ChEBI | | (2S,5R,6R)-6-{[(1-aminocyclohexyl)carbonyl]amino}-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid | ChEBI | | 6-(1-Aminocyclohexanecarboxamido)penicillanic acid | ChEBI | | 6-(1-Aminocyclohexylcarboxamido)penicillanic acid | ChEBI | | Bastcillin | ChEBI | | Calthor | ChEBI | | Ciclacilina | ChEBI | | Ciclacillin | ChEBI | | Ciclacilline | ChEBI | | Ciclacillinum | ChEBI | | Citosarin | ChEBI | | Cyclapen | ChEBI | | Cyclapen-W | ChEBI | | Syngacillin | ChEBI | | Ultracillin | ChEBI | | Vastcillin | ChEBI | | Vipicil | ChEBI | | Wyvital | ChEBI | | ACPC | Kegg | | (2S,5R,6R)-6-{[(1-aminocyclohexyl)carbonyl]amino}-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate | Generator | | 6-(1-Aminocyclohexanecarboxamido)penicillanate | Generator | | 6-(1-Aminocyclohexylcarboxamido)penicillanate | Generator | | Aminocyclohexylpenicillin | HMDB | | Ciclacillum | HMDB |
|
|---|
| Chemical Formula | C15H23N3O4S |
|---|
| Average Molecular Mass | 341.426 g/mol |
|---|
| Monoisotopic Mass | 341.141 g/mol |
|---|
| CAS Registry Number | 3485-14-1 |
|---|
| IUPAC Name | (2S,5R,6R)-6-(1-aminocyclohexaneamido)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid |
|---|
| Traditional Name | cyclacillin |
|---|
| SMILES | [H][C@]12SC(C)(C)[C@@H](N1C(=O)[C@H]2NC(=O)C1(N)CCCCC1)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C15H23N3O4S/c1-14(2)9(12(20)21)18-10(19)8(11(18)23-14)17-13(22)15(16)6-4-3-5-7-15/h8-9,11H,3-7,16H2,1-2H3,(H,17,22)(H,20,21)/t8-,9+,11-/m1/s1 |
|---|
| InChI Key | HGBLNBBNRORJKI-WCABBAIRSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as penicillins. These are organic compounds containing the penicillin core structure, which is structurally characterized by a penam ring bearing two methyl groups at position 2, and an amide group at position 6 [starting from the sulfur atom at position 1]. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Lactams |
|---|
| Sub Class | Beta lactams |
|---|
| Direct Parent | Penicillins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Penicillin
- N-acyl-alpha amino acid or derivatives
- Alpha-amino acid amide
- Alpha-amino acid or derivatives
- Cyclohexylamine
- Thiazolidine
- Tertiary carboxylic acid amide
- Amino acid or derivatives
- Azetidine
- Amino acid
- Carboxamide group
- Secondary carboxylic acid amide
- Carboxylic acid derivative
- Carboxylic acid
- Azacycle
- Monocarboxylic acid or derivatives
- Dialkylthioether
- Hemithioaminal
- Thioether
- Primary aliphatic amine
- Organic oxygen compound
- Organic nitrogen compound
- Hydrocarbon derivative
- Amine
- Organopnictogen compound
- Organonitrogen compound
- Organooxygen compound
- Organic oxide
- Primary amine
- Carbonyl group
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0002-9110000000-880dc522fc28c1bba7cb | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-0002-9000000000-68fd157b54c0f0c04c1a | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03ea-3913000000-e1b225a6069c25031e80 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01pk-6900000000-9015a0229a34616fbc41 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-05u5-9100000000-ebbfbef8f7baae6dadba | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0911000000-0b9e6a3d9d3b19d46cac | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-0930000000-738172cee45fa138ecea | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-002b-9400000000-e5e5efd7b191cd291cea | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0009000000-f94d572cb27dd62c4495 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004l-4529000000-90a2bebebc953a6db79c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a59-7922000000-40fa844297d16d9e505b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0019000000-02832c883fa4bd6dfa39 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0007-9834000000-12493a6db58de235a70b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00di-8901000000-5c2d54fc89ea21fd80a1 | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB01000 |
|---|
| HMDB ID | HMDB0015135 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Ciclacillin |
|---|
| Chemspider ID | 17941 |
|---|
| ChEBI ID | 31444 |
|---|
| PubChem Compound ID | 19003 |
|---|
| Kegg Compound ID | C12766 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|