| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 03:39:14 UTC |
|---|
| Update Date | 2016-11-09 01:15:26 UTC |
|---|
| Accession Number | CHEM016642 |
|---|
| Identification |
|---|
| Common Name | Clothiapine |
|---|
| Class | Small Molecule |
|---|
| Description | Clothiapine has been approved in European countries and is an atypical antipsychotic, shown to be useful in some treatment-resistant patients. |
|---|
| Contaminant Sources | - STOFF IDENT Compounds
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
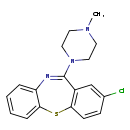
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Clotiapine | Kegg | | Entumin | Kegg | | Entumine | MeSH |
|
|---|
| Chemical Formula | C18H18ClN3S |
|---|
| Average Molecular Mass | 343.870 g/mol |
|---|
| Monoisotopic Mass | 343.091 g/mol |
|---|
| CAS Registry Number | 2058-52-8 |
|---|
| IUPAC Name | 13-chloro-10-(4-methylpiperazin-1-yl)-2-thia-9-azatricyclo[9.4.0.0³,⁸]pentadeca-1(11),3,5,7,9,12,14-heptaene |
|---|
| Traditional Name | dibenzothiazepine |
|---|
| SMILES | CN1CCN(CC1)C1=NC2=CC=CC=C2SC2=C1C=C(Cl)C=C2 |
|---|
| InChI Identifier | InChI=1S/C18H18ClN3S/c1-21-8-10-22(11-9-21)18-14-12-13(19)6-7-16(14)23-17-5-3-2-4-15(17)20-18/h2-7,12H,8-11H2,1H3 |
|---|
| InChI Key | KAAZGXDPUNNEFN-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as dibenzothiazepines. Dibenzothiazepines are compounds containing a dibenzothiazepine moiety, which consists of two benzene connected by a thiazepine ring. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzothiazepines |
|---|
| Sub Class | Dibenzothiazepines |
|---|
| Direct Parent | Dibenzothiazepines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Dibenzothiazepine
- Diarylthioether
- Aryl thioether
- N-alkylpiperazine
- N-methylpiperazine
- Aryl chloride
- Aryl halide
- 1,4-diazinane
- Imidolactam
- Piperazine
- Benzenoid
- Tertiary amine
- Tertiary aliphatic amine
- Amidine
- Carboxylic acid amidine
- Azacycle
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Thioether
- Hydrocarbon derivative
- Organohalogen compound
- Organochloride
- Organonitrogen compound
- Organic nitrogen compound
- Organopnictogen compound
- Amine
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a4l-9046000000-7ef5d280b2c23fac2c09 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0019000000-cb41367a50e508a9d73c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-0029000000-da578a5ae11038615642 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-4960000000-23f3549c77fe64c50503 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0039000000-4b3fdd9c2c210a883410 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0029000000-f2c072b30b8c055f53f4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00di-9630000000-871baf6539bd00f8882d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0009000000-41034cf4cbec70a4531b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-0019000000-03d722ea74cb82cd7c89 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000l-0491000000-51d1cdc8aab197bdf7a6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0009000000-e4f1204a4ccb3048641e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0009000000-c3e297af50c94ed35bff | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-007x-2192000000-4fd4b74cebf9ebc7a02c | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB13256 |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Clotiapine |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 16351 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|