| Synonyms | | Value | Source |
|---|
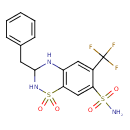
| +--3-Benzyl-3,4-dihydro-6-(trifluoromethyl)-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide | ChEBI | | 6-Trifluoromethyl-3-benzyl-7-sulfamyl-3,4-dihydro-1,2,4-benzothiadiazine 1,1-dioxide | ChEBI | | Bendrofluazide | ChEBI | | Bendroflumethiazidum | ChEBI | | Bendroflumetiazida | ChEBI | | Benzhydroflumethiazide | ChEBI | | Naturetin | Kegg | | +--3-Benzyl-3,4-dihydro-6-(trifluoromethyl)-2H-1,2,4-benzothiadiazine-7-sulphonamide 1,1-dioxide | Generator | | 6-Trifluoromethyl-3-benzyl-7-sulphamyl-3,4-dihydro-1,2,4-benzothiadiazine 1,1-dioxide | Generator | | Bendroflumethazide | HMDB | | Bendrofumethiazide | HMDB | | Benzydroflumethiazide | HMDB | | Benzylhydroflumethiazide | HMDB | | BHFT | HMDB | | Bendroflumethiazide apothecon brand | MeSH, HMDB | | Bristol-myers squibb brand OF bendroflumethiazide | MeSH, HMDB | | DDSA brand OF bendroflumethiazide | MeSH, HMDB | | Esberizid | MeSH, HMDB | | Naturine | MeSH, HMDB | | NeoNaClex | MeSH, HMDB | | Schaper and brümmer brand OF bendroflumethiazide | MeSH, HMDB | | Bendroflumethiazide berk brand | MeSH, HMDB | | Bendroflumethiazide ddsa brand | MeSH, HMDB | | Bendroflumethiazide goldshield brand | MeSH, HMDB | | Bendroflumethiazide leo brand | MeSH, HMDB | | Bristol myers squibb brand OF bendroflumethiazide | MeSH, HMDB | | Urizid | MeSH, HMDB | | Aprinox | MeSH, HMDB | | Bendroflumethiazide protea brand | MeSH, HMDB | | Benzide | MeSH, HMDB | | BenzideM | MeSH, HMDB | | Berkozide | MeSH, HMDB | | leo Brand OF bendroflumethiazide | MeSH, HMDB | | neo NaClex | MeSH, HMDB | | Protea brand OF bendroflumethiazide | MeSH, HMDB | | Apothecon brand OF bendroflumethiazide | MeSH, HMDB | | Bendroflumethiazide knoll brand | MeSH, HMDB | | Benzide m | MeSH, HMDB | | Benzide-m | MeSH, HMDB | | Berk brand OF bendroflumethiazide | MeSH, HMDB | | Centyl | MeSH, HMDB | | Goldshield brand OF bendroflumethiazide | MeSH, HMDB | | Knoll brand OF bendroflumethiazide | MeSH, HMDB | | neo-NaClex | MeSH, HMDB | | Pluryl | MeSH, HMDB |
|
|---|
| InChI Identifier | InChI=1S/C15H14F3N3O4S2/c16-15(17,18)10-7-11-13(8-12(10)26(19,22)23)27(24,25)21-14(20-11)6-9-4-2-1-3-5-9/h1-5,7-8,14,20-21H,6H2,(H2,19,22,23) |
|---|