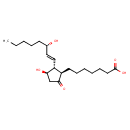
| (11alpha,13E,15S)-11,15-Dihydroxy-9-oxoprost-13-en-1-Oic acid | ChEBI |
| (13E)-(15S)-11alpha,15-Dihydroxy-9-oxoprost-13-enoate | ChEBI |
| 11alpha,15alpha-Dihydroxy-9-oxo-13-trans-prostenoic acid | ChEBI |
| Alprostadil | ChEBI |
| Alprostadilum | ChEBI |
| Befar | ChEBI |
| Caverject | ChEBI |
| Edex | ChEBI |
| Muse | ChEBI |
| PGE-1 | ChEBI |
| PGE1 | ChEBI |
| Prostin VR | ChEBI |
| Prostin VR pediatric | Kegg |
| (11a,13E,15S)-11,15-Dihydroxy-9-oxoprost-13-en-1-Oate | Generator |
| (11a,13E,15S)-11,15-Dihydroxy-9-oxoprost-13-en-1-Oic acid | Generator |
| (11alpha,13E,15S)-11,15-Dihydroxy-9-oxoprost-13-en-1-Oate | Generator |
| (11Α,13E,15S)-11,15-dihydroxy-9-oxoprost-13-en-1-Oate | Generator |
| (11Α,13E,15S)-11,15-dihydroxy-9-oxoprost-13-en-1-Oic acid | Generator |
| (13E)-(15S)-11a,15-Dihydroxy-9-oxoprost-13-enoate | Generator |
| (13E)-(15S)-11a,15-Dihydroxy-9-oxoprost-13-enoic acid | Generator |
| (13E)-(15S)-11alpha,15-Dihydroxy-9-oxoprost-13-enoic acid | Generator |
| (13E)-(15S)-11Α,15-dihydroxy-9-oxoprost-13-enoate | Generator |
| (13E)-(15S)-11Α,15-dihydroxy-9-oxoprost-13-enoic acid | Generator |
| 11a,15a-Dihydroxy-9-oxo-13-trans-prostenoate | Generator |
| 11a,15a-Dihydroxy-9-oxo-13-trans-prostenoic acid | Generator |
| 11alpha,15alpha-Dihydroxy-9-oxo-13-trans-prostenoate | Generator |
| 11Α,15α-dihydroxy-9-oxo-13-trans-prostenoate | Generator |
| 11Α,15α-dihydroxy-9-oxo-13-trans-prostenoic acid | Generator |
| Abbott brand OF alprostadil | MeSH, HMDB |
| Minprog | MeSH, HMDB |
| Paladin brand OF alprostadil | MeSH, HMDB |
| Prostaglandin e1alpha | MeSH, HMDB |
| Schwarz pharma brand OF alprostadil | MeSH, HMDB |
| Astra brand OF alprostadil | MeSH, HMDB |
| Hoyer brand OF alprostadil | MeSH, HMDB |
| Sugiran | MeSH, HMDB |
| Allphar brand OF alprostadil | MeSH, HMDB |
| AstraZeneca brand OF alprostadil | MeSH, HMDB |
| lipo PGE1 | MeSH, HMDB |
| Viridal | MeSH, HMDB |
| lipo-PGE1 | MeSH, HMDB |
| Vasaprostan | MeSH, HMDB |
| Janssen brand OF alprostadil | MeSH, HMDB |
| PGE1alpha | MeSH, HMDB |
| Pharmacia brand 1 OF alprostadil | MeSH, HMDB |
| Prostavasin | MeSH, HMDB |
| Prostine VR | MeSH, HMDB |
| Pharmacia brand 2 OF alprostadil | MeSH, HMDB |
| Prostaglandin e1 | MeSH, KEGG |
| Schwarz brand OF alprostadil | MeSH, HMDB |
| Vivus brand OF alprostadil | MeSH, HMDB |
| Prink | KEGG, HMDB |
| Vitaros | KEGG, HMDB |
| Caverject impulse | ChEMBL, HMDB |
| U-10136prostin | ChEMBL, HMDB |
| U-10136alprostadil | ChEMBL, HMDB |
| (+)-3-Hydroxy-2-(3-hydroxy-1-octenyl)-5-oxo-cyclopentaneheptanoate | HMDB |
| (+)-3-Hydroxy-2-(3-hydroxy-1-octenyl)-5-oxo-cyclopentaneheptanoic acid | HMDB |
| (-)-3-Hydroxy-2-(3-hydroxy-1-octenyl)-5-oxo-cyclopentaneheptanoate | HMDB |
| (-)-3-Hydroxy-2-(3-hydroxy-1-octenyl)-5-oxo-cyclopentaneheptanoic acid | HMDB |
| (-)-Prostaglandin e1 | HMDB |
| (13E)-(15S)-11,15-Dihydroxy-9-oxoprost-13-enoate | HMDB |
| (13E)-(15S)-11,15-Dihydroxy-9-oxoprost-13-enoic acid | HMDB |
| (13E)-(15S)-11-alpha,15-Dihydroxy-9-oxoprost-13-enoate | HMDB |
| (13E)-(15S)-11-alpha,15-Dihydroxy-9-oxoprost-13-enoic acid | HMDB |
| 11,15-Dihydroxy-9-oxoprost-13-en-1-Oate | HMDB |
| 11,15-Dihydroxy-9-oxoprost-13-en-1-Oic acid | HMDB |
| 11,15-Dihydroxy-9-oxoprost-13-en-1-Oic acid (acd/name 4.0) | HMDB |
| 3-Hydroxy-2-(3-hydroxy-1-octenyl)-5-oxo-cyclopentaneheptanoate | HMDB |
| 3-Hydroxy-2-(3-hydroxy-1-octenyl)-5-oxo-cyclopentaneheptanoic acid | HMDB |
| Alprostadil prostoglandin e1 | HMDB |
| Alprostadil(usan) | HMDB |
| L-Prostaglandin e1 | HMDB |