| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-20 16:17:12 UTC |
|---|
| Update Date | 2016-11-09 01:15:20 UTC |
|---|
| Accession Number | CHEM016157 |
|---|
| Identification |
|---|
| Common Name | Symphytine |
|---|
| Class | Small Molecule |
|---|
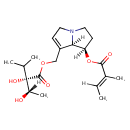
| Description | A but-2-enoate ester obtained by formal condensation of the carboxy group of (2E)-2-methylbut-2-enoic acid with the 1-hydroxy group of (1R,7aR)-7-({oxy}methyl)-2,3,5,7a-tetrahydro-1H-pyrrolizin-1-ol. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 7-Tiglyl-9-(-)-viridiflorylretronecine | ChEBI | | 7-Tiglylretronecine viridiflorate | ChEBI | | 7-Tiglylretronecine viridifloric acid | Generator | | [(7R,8R)-7-[(e)-2-Methylbut-2-enoyl]oxy-5,6,7,8-tetrahydro-3H-pyrrolizin-1-yl]methyl (2S)-2-hydroxy-2-[(1S)-1-hydroxyethyl]-3-methylbutanoic acid | Generator | | 7-Tiglylretronecine viridiflate | Generator | | 7-Tiglylretronecine viridiflic acid | Generator | | Symphytine | MeSH |
|
|---|
| Chemical Formula | C20H31NO6 |
|---|
| Average Molecular Mass | 381.469 g/mol |
|---|
| Monoisotopic Mass | 381.215 g/mol |
|---|
| CAS Registry Number | 22571-95-5 |
|---|
| IUPAC Name | (1R,7aR)-7-({[(2S)-2-hydroxy-2-[(1S)-1-hydroxyethyl]-3-methylbutanoyl]oxy}methyl)-2,3,5,7a-tetrahydro-1H-pyrrolizin-1-yl (2E)-2-methylbut-2-enoate |
|---|
| Traditional Name | symphytine |
|---|
| SMILES | [H]\C(C)=C(\C)C(=O)O[C@]1([H])CCN2CC=C(COC(=O)[C@](O)(C(C)C)[C@]([H])(C)O)[C@]12[H] |
|---|
| InChI Identifier | InChI=1S/C20H31NO6/c1-6-13(4)18(23)27-16-8-10-21-9-7-15(17(16)21)11-26-19(24)20(25,12(2)3)14(5)22/h6-7,12,14,16-17,22,25H,8-11H2,1-5H3/b13-6+/t14-,16+,17+,20-/m0/s1 |
|---|
| InChI Key | MVWPTZQHBOWRTF-SMLWLWDZSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as alkaloids and derivatives. These are naturally occurring chemical compounds that contain mostly basic nitrogen atoms. This group also includes some related compounds with neutral and even weakly acidic properties. Also some synthetic compounds of similar structure are attributed to alkaloids. In addition to carbon, hydrogen and nitrogen, alkaloids may also contain oxygen, sulfur and more rarely other elements such as chlorine, bromine, and phosphorus. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Alkaloids and derivatives |
|---|
| Class | Not Available |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Alkaloids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alkaloid or derivatives
- Pyrrolizine
- Beta-hydroxy acid
- Fatty acid ester
- Dicarboxylic acid or derivatives
- Hydroxy acid
- Fatty acyl
- N-alkylpyrrolidine
- Pyrrolidine
- Pyrroline
- Alpha,beta-unsaturated carboxylic ester
- Enoate ester
- Tertiary alcohol
- 1,2-diol
- Amino acid or derivatives
- Carboxylic acid ester
- Secondary alcohol
- Tertiary aliphatic amine
- Tertiary amine
- Carboxylic acid derivative
- Azacycle
- Organoheterocyclic compound
- Organooxygen compound
- Organonitrogen compound
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Carbonyl group
- Hydrocarbon derivative
- Amine
- Alcohol
- Organic nitrogen compound
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-4479000000-5217b0e8be9a2e358b45 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-008d-9440000000-5cd3a0138e10c50ca139 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-001j-9700000000-5f2eb08f8b9a61307483 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00m0-2529000000-664bae0257eeb2396484 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-07cm-4945000000-2e6d7cd8a3a1f3025b72 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01b9-7900000000-e77cf13f42074cc0c790 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00002123 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 9375 |
|---|
| PubChem Compound ID | 5281754 |
|---|
| Kegg Compound ID | C10409 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|