| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-20 16:15:37 UTC |
|---|
| Update Date | 2016-11-09 01:15:20 UTC |
|---|
| Accession Number | CHEM016123 |
|---|
| Identification |
|---|
| Common Name | Light Green SF |
|---|
| Class | Small Molecule |
|---|
| Description | An organic sodium salt having 3-amino}phenyl)(4-sulfonatophenyl)methylidene]cyclohexa-2,5-dien-1-ylidene}azaniumyl)methyl]benzene-1-sulfonate as the counterion. It is the standard dye in North American for staining collagen and is also used extensively in plant histology. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
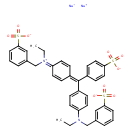
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| a F Green no.2 | ChEBI | | A.F. green no. 2 | ChEBI | | Acid brilliant green SF | ChEBI | | Acid green | ChEBI | | Acid green a | ChEBI | | Acidal light green SF | ChEBI | | Acilan green SFG | ChEBI | | Amacid green g | ChEBI | | C.I. 42095 | ChEBI | | C.I. 670 | ChEBI | | C.I. acid green 5 | ChEBI | | C.I. acid green 5, disodium salt | ChEBI | | C.I. FOOD green 2 | ChEBI | | CCRIS 889 | ChEBI | | D And C green no. 4 | ChEBI | | D&C green no. 2 | ChEBI | | Fast acid green N | ChEBI | | FD And C green no. 2 | ChEBI | | FD And C green no. 4 | ChEBI | | Fenazo green 7g | ChEBI | | FOOD Green 2 | ChEBI | | Green no. 203 | ChEBI | | Leather green SF | ChEBI | | Lichtgruen | ChEBI | | Light green | ChEBI | | Light green 2GN | ChEBI | | Light green CF | ChEBI | | Light green FCF yellowish | ChEBI | | Light green FS | ChEBI | | Light green g | ChEBI | | Light green lake | ChEBI | | Light green S | ChEBI | | Light green SF | ChEBI | | Light green sfa | ChEBI | | Light green SFD | ChEBI | | Light green yellowish | ChEBI | | Light SF yellowish (biological stain) | ChEBI | | Lissamine green SF | ChEBI | | Lissamine lake green SF | ChEBI | | Merantine green SF | ChEBI | | MY/68 | ChEBI | | NSC 9619 | ChEBI | | Pencil green SF | ChEBI | | SulfO green J | ChEBI | | Sumitomo light green SF yellowish | ChEBI | | Wool brilliant green SF | ChEBI | | SulphO green J | Generator | | Light green SF yellowish | ChEBI |
|
|---|
| Chemical Formula | C37H34N2Na2O9S3 |
|---|
| Average Molecular Mass | 792.848 g/mol |
|---|
| Monoisotopic Mass | 792.122 g/mol |
|---|
| CAS Registry Number | 5141-20-8 |
|---|
| IUPAC Name | disodium 3-{[ethyl({4-[(4-{ethyl[(3-sulfonatophenyl)methyl]iminiumyl}cyclohexa-2,5-dien-1-ylidene)(4-sulfonatophenyl)methyl]phenyl})amino]methyl}benzene-1-sulfonate |
|---|
| Traditional Name | disodium 3-{[ethyl({4-[(4-{ethyl[(3-sulfonatophenyl)methyl]iminio}cyclohexa-2,5-dien-1-ylidene)(4-sulfonatophenyl)methyl]phenyl})amino]methyl}benzenesulfonate |
|---|
| SMILES | [Na+].[Na+].CCN(CC1=CC(=CC=C1)S([O-])(=O)=O)C1=CC=C(C=C1)C(C1=CC=C(C=C1)S([O-])(=O)=O)=C1C=CC(C=C1)=[N+](CC)CC1=CC=CC(=C1)S([O-])(=O)=O |
|---|
| InChI Identifier | InChI=1S/C37H36N2O9S3.2Na/c1-3-38(25-27-7-5-9-35(23-27)50(43,44)45)32-17-11-29(12-18-32)37(31-15-21-34(22-16-31)49(40,41)42)30-13-19-33(20-14-30)39(4-2)26-28-8-6-10-36(24-28)51(46,47)48;;/h5-24H,3-4,25-26H2,1-2H3,(H2-,40,41,42,43,44,45,46,47,48);;/q;2*+1/p-2 |
|---|
| InChI Key | DGOBMKYRQHEFGQ-UHFFFAOYSA-L |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenylbenzamines. These are aromatic compounds consisting of a benzyl group that is N-linked to a benzamine. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Phenylmethylamines |
|---|
| Direct Parent | Phenylbenzamines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenylbenzamine
- Diphenylmethane
- Benzenesulfonate
- Arylsulfonic acid or derivatives
- Benzenesulfonyl group
- 1-sulfo,2-unsubstituted aromatic compound
- Benzylamine
- Tertiary aliphatic/aromatic amine
- Aniline or substituted anilines
- Dialkylarylamine
- Aralkylamine
- Azomethine
- Secondary ketimine
- Organic sulfonic acid or derivatives
- Organosulfonic acid or derivatives
- Organosulfonic acid
- Sulfonyl
- Tertiary amine
- Organic alkali metal salt
- Propargyl-type 1,3-dipolar organic compound
- Organic 1,3-dipolar compound
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Amine
- Organic nitrogen compound
- Organosulfur compound
- Organonitrogen compound
- Hydrocarbon derivative
- Organic salt
- Organic sodium salt
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-000l-1000019300-04b04153c907756291f3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0000001900-4d51bfe5f098087b53b7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000l-0000119300-13d1afb671f6ee91699a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01p9-0000009000-e6e60bcff14fa5906766 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0000000900-4bfed5a5a0688091a220 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0000000900-4bfed5a5a0688091a220 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-0000000900-4bfed5a5a0688091a220 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB11183 |
|---|
| HMDB ID | HMDB0032282 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 87065 |
|---|
| PubChem Compound ID | 21223 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|