| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-20 16:14:47 UTC |
|---|
| Update Date | 2016-11-09 01:15:20 UTC |
|---|
| Accession Number | CHEM016101 |
|---|
| Identification |
|---|
| Common Name | Vincristine sulfate |
|---|
| Class | Small Molecule |
|---|
| Description | An organic sulfate salt containing equimolar amounts of vincristine(2+) and sulfate. Used for the treatment of a variety of cancers. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
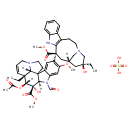
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Marqibo | ChEBI | | Oncovin | ChEBI | | Vincasar | ChEBI | | Vincrex | ChEBI | | VCR | Kegg | | Vincristine sulfuric acid | Generator | | Vincristine sulphate | Generator | | Vincristine sulphuric acid | Generator | | Onkocristin | MeSH | | Leurocristine | MeSH | | Vincristine | MeSH | | Vincristine sulfate | MeSH | | Sulfate, vincristine | MeSH | | Vincasar PFS | MeSH | | Vincrisul | MeSH | | Farmistin | MeSH | | Oncovine | MeSH | | PFS, Vincasar | MeSH | | Vincristin medac | MeSH | | Cellcristin | MeSH | | Vincristin bristol | MeSH | | Vintec | MeSH | | Citomid | MeSH | | Sulfate | | | vincristine | | | Sulphate | | | Sulphuric acid | |
|
|---|
| Chemical Formula | C46H58N4O14S |
|---|
| Average Molecular Mass | 923.040 g/mol |
|---|
| Monoisotopic Mass | 922.367 g/mol |
|---|
| CAS Registry Number | 2068-78-2 |
|---|
| IUPAC Name | methyl (1R,9R,10S,11R,12R,19R)-11-(acetyloxy)-12-ethyl-4-[(13S,15S,17S)-17-ethyl-17-hydroxy-13-(methoxycarbonyl)-1,11-diazatetracyclo[13.3.1.0⁴,¹².0⁵,¹⁰]nonadeca-4(12),5,7,9-tetraen-13-yl]-8-formyl-10-hydroxy-5-methoxy-8,16-diazapentacyclo[10.6.1.0¹,⁹.0²,⁷.0¹⁶,¹⁹]nonadeca-2,4,6,13-tetraene-10-carboxylate; sulfuric acid |
|---|
| Traditional Name | sulfuric acid; vincristine |
|---|
| SMILES | OS(O)(=O)=O.[H][C@@]12N3CC[C@@]11C4=CC(=C(OC)C=C4N(C=O)[C@@]1([H])[C@@](O)(C(=O)OC)[C@]([H])(OC(C)=O)[C@]2(CC)C=CC3)[C@]1(C[C@]2([H])CN(C[C@](O)(CC)C2)CCC2=C1NC1=CC=CC=C21)C(=O)OC |
|---|
| InChI Identifier | InChI=1S/C46H56N4O10.H2O4S/c1-7-42(55)22-28-23-45(40(53)58-5,36-30(14-18-48(24-28)25-42)29-12-9-10-13-33(29)47-36)32-20-31-34(21-35(32)57-4)50(26-51)38-44(31)16-19-49-17-11-15-43(8-2,37(44)49)39(60-27(3)52)46(38,56)41(54)59-6;1-5(2,3)4/h9-13,15,20-21,26,28,37-39,47,55-56H,7-8,14,16-19,22-25H2,1-6H3;(H2,1,2,3,4)/t28-,37+,38-,39-,42+,43-,44-,45+,46+;/m1./s1 |
|---|
| InChI Key | AQTQHPDCURKLKT-JKDPCDLQSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as oxanes. Oxanes are compounds containing an oxane (tetrahydropyran) ring, which is a six-member saturated aliphatic heterocycle with one oxygen atom and five carbon atoms. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Oxanes |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Oxanes |
|---|
| Alternative Parents | |
|---|
| Substituents | - Oxane
- Oxacycle
- Ether
- Oxirane
- Dialkyl ether
- Organic oxygen compound
- Hydrocarbon derivative
- Organooxygen compound
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-0000000009-00e5ce550ad91f29c0d3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00di-0000000009-00e5ce550ad91f29c0d3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00di-0000000009-00e5ce550ad91f29c0d3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0000000009-cac82c0fed91d632b050 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-0000000009-cac82c0fed91d632b050 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00di-0000000009-cac82c0fed91d632b050 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Vincristine |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 79401 |
|---|
| PubChem Compound ID | 249332 |
|---|
| Kegg Compound ID | C13974 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|