| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-20 16:13:07 UTC |
|---|
| Update Date | 2016-11-09 01:15:19 UTC |
|---|
| Accession Number | CHEM016065 |
|---|
| Identification |
|---|
| Common Name | Retrorsine |
|---|
| Class | Small Molecule |
|---|
| Description | |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
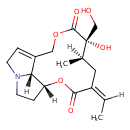
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Retrorsine hydrochloride | MeSH | | Usaramine | MeSH | | Retrorsine, (15E)-isomer | MeSH |
|
|---|
| Chemical Formula | C18H25NO6 |
|---|
| Average Molecular Mass | 351.399 g/mol |
|---|
| Monoisotopic Mass | 351.168 g/mol |
|---|
| CAS Registry Number | 480-54-6 |
|---|
| IUPAC Name | (1R,4Z,6R,7S,17R)-4-ethylidene-7-hydroxy-7-(hydroxymethyl)-6-methyl-2,9-dioxa-14-azatricyclo[9.5.1.0¹⁴,¹⁷]heptadec-11-ene-3,8-dione |
|---|
| Traditional Name | retrorsine |
|---|
| SMILES | [H]\C(C)=C1/C[C@@]([H])(C)[C@](O)(CO)C(=O)OCC2=CCN3CC[C@@]([H])(OC1=O)[C@@]23[H] |
|---|
| InChI Identifier | InChI=1S/C18H25NO6/c1-3-12-8-11(2)18(23,10-20)17(22)24-9-13-4-6-19-7-5-14(15(13)19)25-16(12)21/h3-4,11,14-15,20,23H,5-10H2,1-2H3/b12-3-/t11-,14-,15-,18-/m1/s1 |
|---|
| InChI Key | BCJMNZRQJAVDLD-CQRYIUNCSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as macrolides and analogues. These are organic compounds containing a lactone ring of at least twelve members. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Macrolides and analogues |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Macrolides and analogues |
|---|
| Alternative Parents | |
|---|
| Substituents | - Senecionan-skeleton
- Macrolide
- Alkaloid or derivatives
- Pyrrolizine
- Dicarboxylic acid or derivatives
- N-alkylpyrrolidine
- Pyrrolidine
- Pyrroline
- Tertiary alcohol
- Alpha,beta-unsaturated carboxylic ester
- Enoate ester
- 1,2-diol
- Amino acid or derivatives
- Carboxylic acid ester
- Lactone
- Tertiary amine
- Tertiary aliphatic amine
- Carboxylic acid derivative
- Oxacycle
- Azacycle
- Organoheterocyclic compound
- Organic oxide
- Primary alcohol
- Organooxygen compound
- Organonitrogen compound
- Organopnictogen compound
- Organic nitrogen compound
- Organic oxygen compound
- Carbonyl group
- Hydrocarbon derivative
- Amine
- Alcohol
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0619000000-2b2d5bb8fba3d0d2ba77 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0fer-3914000000-47baf7858c4d59d81028 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-062i-3900000000-8fb8b06d63ab8506063c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0f79-0829000000-54670a883add739fdd4d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-059i-0903000000-4b9d9418e60d6e922065 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052f-9600000000-651f40ea07bc8cd43fe4 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00002109 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 5281743 |
|---|
| Kegg Compound ID | C10364 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|