| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-19 04:33:17 UTC |
|---|
| Update Date | 2016-11-09 01:15:02 UTC |
|---|
| Accession Number | CHEM014622 |
|---|
| Identification |
|---|
| Common Name | Ethanol, 2,2'-oxybis-, 1,1'-dinitrate |
|---|
| Class | Small Molecule |
|---|
| Description | Diethylene glycol dinitrate is a nitrated alcohol ester produced by the action of concentrated nitric acid, normally admixed with an excess of strong sulfuric acid as a dehydrating agent, upon diethylene glycol.
Diethylene glycol dinitrate is a colorless, odorless, viscous, oily liquid, with specific gravity 1.4092 at 0 °C and 1.3846 at 20 °C; freezing point −11.5 °C under a standard atmosphere; the theoretical boiling point of approximately 197 °C difficult to confirm as the compound begins to decompose and spontaneously inflames at or slightly below this temperature. Partial pressure is reported as 0.007 mmHg (930 mPa) at 22.4 °C and 760 mmHg (101 kPa). It is readily miscible in most non-polar solvents, methanol, and cold acetic acid. Solubility in water (4.1 g/L at 24 °C) and ethanol is very low. While chemically similar to a number of powerful high explosives, pure diethylene glycol dinitrate is extremely hard to initiate and will not propagate a detonation wave. It inflames only with difficulty (requiring localized heating to decomposition point) unless first atomized, and burns placidly even in quantity.
Mixed with nitrocellulose and extruded under pressure, diethylene glycol dinitrate forms a tough colloid whose characteristics (good specific impulse, moderate burn rate and temperature, great resistance to accidental ignition and casual handling) make it well suited as a smokeless powder for artillery and a solid propellant for rocketry. It was widely used in this capacity during World War II (e.g., by the Kriegsmarine). It also found use as a "productive" desensitizer (one that contributes to the overall power of the explosion rather than having a neutral or negative effect) in nitroglycerine and nitroglycol-based explosives such as dynamite and blasting gelatin. It is also used as plasticizer for energetic materials.
If ingested, like nitroglycerine, it produces rapid vasodilation through the release of nitrogen monoxide, NO. Popularly termed nitric oxide, NO is a physiological signaling molecule that relaxes smooth muscle. Consequently, diethylene glycol dinitrate has occasionally been used medically to relieve angina, substernal chest pain associated with impaired cardiac circulation. The rationale is that the concurrent headache it induces is somewhat less severe than other nitro compounds.
At present, interest in the chemical seems to be mostly historical: more potent perchlorate–metal mixtures have long since supplanted it as a solid propellant; safer explosives have replaced nitroglycerine, true dynamites (the term is often used generically, even by experienced field technicians, to refer to almost any explosive supplied in small, discrete packages) retaining only a few specialist uses. The medical application was never widespread, the standard nitroglycerine being faster acting and very inexpensive; oral nitrates in any case being only palliative, not an effective treatment.
Triethylene glycol dinitrate, diethylene glycol dinitrate, and trimethylolethane trinitrate are being considered as replacements for nitroglycerin in propellants. |
|---|
| Contaminant Sources | - HPV EPA Chemicals
- STOFF IDENT Compounds
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Diethylene glycol dinitric acid | Generator | | 2-[2-(Nitrooxy)ethoxy]ethyl nitric acid | Generator |
|
|---|
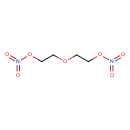
| Chemical Formula | C4H8N2O7 |
|---|
| Average Molecular Mass | 196.115 g/mol |
|---|
| Monoisotopic Mass | 196.033 g/mol |
|---|
| CAS Registry Number | 693-21-0 |
|---|
| IUPAC Name | 2-[2-(nitrooxy)ethoxy]ethyl nitrate |
|---|
| Traditional Name | diethylene glycol dinitrate |
|---|
| SMILES | O=N(=O)OCCOCCON(=O)=O |
|---|
| InChI Identifier | InChI=1S/C4H8N2O7/c7-5(8)12-3-1-11-2-4-13-6(9)10/h1-4H2 |
|---|
| InChI Key | LYAGTVMJGHTIDH-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as alkyl nitrates. These are organic compounds containing a nitrate that is O-linked to an alkyl group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organic oxoanionic compounds |
|---|
| Sub Class | Organic nitrates |
|---|
| Direct Parent | Alkyl nitrates |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alkyl nitrate
- Organic nitro compound
- Organic nitric acid or derivatives
- Organic 1,3-dipolar compound
- Allyl-type 1,3-dipolar organic compound
- Ether
- Dialkyl ether
- Organic nitrogen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0900000000-5b10863e54112631d395 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001j-5900000000-d7927fe11716f41f7073 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-002e-9000000000-b24790cec7745ac97886 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0900000000-d1a973223c3b774282cc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-2900000000-1a16f0ba270662f1f9c4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4r-9400000000-873a61aa8de0e3bd4bc3 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Diethylene glycol dinitrate |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 61198 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|