| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-19 04:00:42 UTC |
|---|
| Update Date | 2016-11-09 01:14:38 UTC |
|---|
| Accession Number | CHEM012715 |
|---|
| Identification |
|---|
| Common Name | Acetic acid, mercapto-, monoammonium salt |
|---|
| Class | Small Molecule |
|---|
| Description | |
|---|
| Contaminant Sources | - HPV EPA Chemicals
- OECD HPV Chemicals
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| AMMONIUM thioglycolic acid | Generator | | 2-Mercaptoacetate, calcium salt (2:1) salt, trihydrate | MeSH | | 2-Mercaptoacetate, monopotassium salt | MeSH | | 2-Mercaptoacetate, monosodium salt | MeSH | | Calcium thioglycolate | MeSH | | 2-Mercaptoacetate, monoammonium salt | MeSH | | 2-Mercaptoacetate, bismuth (3+), sodium salt (3:1:3) | MeSH | | Mercaptoacetic acid | MeSH | | 2-Mercaptoacetate | MeSH | | 2-Mercaptoacetate, calcium salt (1:1) | MeSH | | Sodium thioglycollate | MeSH | | 2-Thioglycolic acid | MeSH | | Sodium thioglycolate | MeSH | | 2-Mercaptoacetate, calcium salt (2:1) | MeSH | | Thioglycolic acid | MeSH | | Ammonium thioglycolate | MeSH |
|
|---|
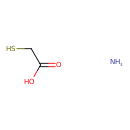
| Chemical Formula | C2H7NO2S |
|---|
| Average Molecular Mass | 109.140 g/mol |
|---|
| Monoisotopic Mass | 109.020 g/mol |
|---|
| CAS Registry Number | 5421-46-5 |
|---|
| IUPAC Name | 2-sulfanylacetic acid amine |
|---|
| Traditional Name | thioglycolic acid amine |
|---|
| SMILES | N.OC(=O)CS |
|---|
| InChI Identifier | InChI=1S/C2H4O2S.H3N/c3-2(4)1-5;/h5H,1H2,(H,3,4);1H3 |
|---|
| InChI Key | ZZTCCAPMZLDHFM-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as alpha-mercaptocarboxylic acids. These are carboxylic acids that bear a thiol group at the C-2 position. Alpha-mercaptocarboxylic acids have the general formula RC(S)C(=O)O, where R = H, organyl group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Carboxylic acids |
|---|
| Direct Parent | alpha-Mercaptocarboxylic acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - 2-mercaptocarboxylic acid
- Carboxylic acid salt
- Monocarboxylic acid or derivatives
- Alkylthiol
- Organic nitrogen compound
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organic salt
- Organosulfur compound
- Organooxygen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0900000000-df50415f219fb850f482 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-0900000000-df50415f219fb850f482 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03di-0900000000-df50415f219fb850f482 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0900000000-014afedd22c20457bee7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-0900000000-014afedd22c20457bee7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-0900000000-014afedd22c20457bee7 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 21534 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|