| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-19 03:13:24 UTC |
|---|
| Update Date | 2016-11-09 01:14:06 UTC |
|---|
| Accession Number | CHEM009920 |
|---|
| Identification |
|---|
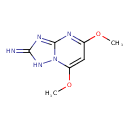
| Common Name | 5,7-Dimethoxy-[1,2,4]triazolo[1,5-a]pyrimidin-2-amine |
|---|
| Class | Small Molecule |
|---|
| Description | |
|---|
| Contaminant Sources | - HPV EPA Chemicals
- STOFF IDENT Compounds
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C7H9N5O2 |
|---|
| Average Molecular Mass | 195.182 g/mol |
|---|
| Monoisotopic Mass | 195.076 g/mol |
|---|
| CAS Registry Number | 13223-43-3 |
|---|
| IUPAC Name | 5,7-dimethoxy-1H,2H-[1,2,4]triazolo[1,5-a]pyrimidin-2-imine |
|---|
| Traditional Name | 5,7-dimethoxy-1H-[1,2,4]triazolo[1,5-a]pyrimidin-2-imine |
|---|
| SMILES | COC1=NC2=NC(=N)NN2C(OC)=C1 |
|---|
| InChI Identifier | InChI=1S/C7H9N5O2/c1-13-4-3-5(14-2)12-7(9-4)10-6(8)11-12/h3H,1-2H3,(H2,8,11) |
|---|
| InChI Key | SJEIPAUSEWIHJI-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as triazolopyrimidines. These are polycyclic aromatic compounds containing triazole ring fused to a pyrimidine ring. Triazole is a five-membered ring consisting of two carbon atoms and three nitrogen atoms. Pyrimidine is a 6-membered ring consisting of four carbon atoms and two nitrogen centers at the 1- and 3- ring positions. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Triazolopyrimidines |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Triazolopyrimidines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Triazolopyrimidine
- Alkyl aryl ether
- Pyrimidine
- Azole
- Triazole
- 1,2,4-triazole
- Heteroaromatic compound
- Ether
- Azacycle
- Amine
- Organic oxygen compound
- Primary amine
- Organooxygen compound
- Organonitrogen compound
- Organopnictogen compound
- Organic nitrogen compound
- Hydrocarbon derivative
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0900000000-0d004afce75e43986246 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03dj-0900000000-1a35419bc8c5e70bfb17 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-001i-9300000000-8ce896bec6c31ecd40ca | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0900000000-4f35e088e0fd90f94280 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-9500000000-b10996e92cd091388ca0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9000000000-1102e470822db3a91e4c | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 11194901 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|