| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-19 02:42:45 UTC |
|---|
| Update Date | 2026-03-26 18:33:49 UTC |
|---|
| Accession Number | CHEM008284 |
|---|
| Identification |
|---|
| Common Name | Metrafenone |
|---|
| Class | Small Molecule |
|---|
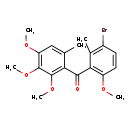
| Description | A member of the class of benzophenones that is benzophenone in which one of the phenyl groups is substituted by methoxy groups at positions 2, 3, and 4 and by a methyl group at position 6, while the other is substituted at positions 2, 3, and 6 by methyl, bromine, and methoxy groups, respectively. A fungicide with protectant and curative properties, it is used for the control of powdery mildew in cereals and grape vines. |
|---|
| Contaminant Sources | - My Exposome Chemicals
- STOFF IDENT Compounds
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3'-Bromo-2,3,4,6'-tetramethoxy-2',6-dimethylbenzophenone | ChEBI | | BAS 560 02F | ChEBI | | Metrafenone | MeSH |
|
|---|
| Chemical Formula | C19H21BrO5 |
|---|
| Average Molecular Mass | 409.276 g/mol |
|---|
| Monoisotopic Mass | 408.057 g/mol |
|---|
| CAS Registry Number | 220899-03-6 |
|---|
| IUPAC Name | (3-bromo-6-methoxy-2-methylphenyl)(2,3,4-trimethoxy-6-methylphenyl)methanone |
|---|
| Traditional Name | (3-bromo-6-methoxy-2-methylphenyl)(2,3,4-trimethoxy-6-methylphenyl)methanone |
|---|
| SMILES | COC1=C(C(=O)C2=C(OC)C(OC)=C(OC)C=C2C)C(C)=C(Br)C=C1 |
|---|
| InChI Identifier | InChI=1S/C19H21BrO5/c1-10-9-14(23-4)18(24-5)19(25-6)15(10)17(21)16-11(2)12(20)7-8-13(16)22-3/h7-9H,1-6H3 |
|---|
| InChI Key | AMSPWOYQQAWRRM-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as benzophenones. These are organic compounds containing a ketone attached to two phenyl groups. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Benzophenones |
|---|
| Direct Parent | Benzophenones |
|---|
| Alternative Parents | |
|---|
| Substituents | - Benzophenone
- Diphenylmethane
- Aryl-phenylketone
- Anisole
- Phenoxy compound
- Benzoyl
- Phenol ether
- Aryl ketone
- Methoxybenzene
- Halobenzene
- Bromobenzene
- Toluene
- Alkyl aryl ether
- Aryl bromide
- Aryl halide
- Ketone
- Ether
- Organooxygen compound
- Organobromide
- Organohalogen compound
- Organic oxygen compound
- Hydrocarbon derivative
- Organic oxide
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0062900000-12907a2418f53b5fd96b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a6r-0091100000-d4b35a8e25baba7f4631 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004i-0960000000-fca5978b2906db43ed2b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0100900000-8c4ef21235e67258a76a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-0526900000-9e471cebb60e5bc9952c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0aos-2912000000-ff087469812d8b6b2c2c | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 83345 |
|---|
| PubChem Compound ID | 6451057 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|