| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-19 02:37:47 UTC |
|---|
| Update Date | 2016-11-09 01:13:43 UTC |
|---|
| Accession Number | CHEM008088 |
|---|
| Identification |
|---|
| Common Name | Uniconizole-P |
|---|
| Class | Small Molecule |
|---|
| Description | A (1E)-1-(4-chlorophenyl)-4,4-dimethyl-2-(1H-1,2,4-triazol-1-yl)pent-1-en-3-ol that has S configuration at the chiral centre. It is the enantiomer of (R)-uniconazole; the fungicide and plant growth retardant uniconazole is the racemic mixture comprising (R)-uniconazole and uniconazole-P. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
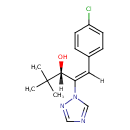
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (AlphaSS,betae)-beta-[(4-chlorophenyl)methylene]-alpha-(1,1-dimethylethyl)-1H-1,2,4-triazole-1-ethanol | ChEBI | | (S)-(+)-Uniconazole | ChEBI | | (S)-Uniconazole | ChEBI | | Lomica | ChEBI | | (AlphaSS,betae)-b-[(4-chlorophenyl)methylene]-a-(1,1-dimethylethyl)-1H-1,2,4-triazole-1-ethanol | Generator | | (AlphaSS,betae)-β-[(4-chlorophenyl)methylene]-α-(1,1-dimethylethyl)-1H-1,2,4-triazole-1-ethanol | Generator | | (S)-e-1-(4-Chlorophenyl)-4,4-dimethyl-2-(1,2,4-triazole-1-yl)penten-3-ol | MeSH | | Uniconazole-P | MeSH |
|
|---|
| Chemical Formula | C15H18ClN3O |
|---|
| Average Molecular Mass | 291.780 g/mol |
|---|
| Monoisotopic Mass | 291.114 g/mol |
|---|
| CAS Registry Number | 83657-17-4 |
|---|
| IUPAC Name | (1E,3S)-1-(4-chlorophenyl)-4,4-dimethyl-2-(1H-1,2,4-triazol-1-yl)pent-1-en-3-ol |
|---|
| Traditional Name | (1E,3S)-1-(4-chlorophenyl)-4,4-dimethyl-2-(1,2,4-triazol-1-yl)pent-1-en-3-ol |
|---|
| SMILES | [H]\C(=C(/N1C=NC=N1)[C@@]([H])(O)C(C)(C)C)C1=CC=C(Cl)C=C1 |
|---|
| InChI Identifier | InChI=1S/C15H18ClN3O/c1-15(2,3)14(20)13(19-10-17-9-18-19)8-11-4-6-12(16)7-5-11/h4-10,14,20H,1-3H3/b13-8+/t14-/m1/s1 |
|---|
| InChI Key | YNWVFADWVLCOPU-MAUPQMMJSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as cinnamyl alcohols. These are aromatic alcohols containing a 3-phenylprop-2-en-1-ol moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Cinnamyl alcohols |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Cinnamyl alcohols |
|---|
| Alternative Parents | |
|---|
| Substituents | - Cinnamyl alcohol
- Chlorobenzene
- Halobenzene
- Aryl chloride
- Aryl halide
- Monocyclic benzene moiety
- Benzenoid
- Azole
- Heteroaromatic compound
- 1,2,4-triazole
- Secondary alcohol
- Azacycle
- Organoheterocyclic compound
- Organohalogen compound
- Organooxygen compound
- Alcohol
- Hydrocarbon derivative
- Organopnictogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Organochloride
- Organonitrogen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0090000000-c9c4c28887fe02fe2e69 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udr-4090000000-b6ed44b5e791189765e4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00di-9120000000-4e57eaa8c6be1a5893b9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-2090000000-c492da33614fae53811a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014l-8090000000-3ad3ab224ecec2e939d6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-014l-9000000000-ff66b2d3807823be1d8b | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 81785 |
|---|
| PubChem Compound ID | 6436639 |
|---|
| Kegg Compound ID | C18494 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|