| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-19 02:34:35 UTC |
|---|
| Update Date | 2016-11-09 01:13:42 UTC |
|---|
| Accession Number | CHEM007971 |
|---|
| Identification |
|---|
| Common Name | Omethoate |
|---|
| Class | Small Molecule |
|---|
| Description | |
|---|
| Contaminant Sources | - My Exposome Chemicals
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
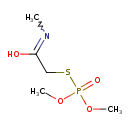
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-Dimethoxyphosphinoylthio-N-methylacetamide | ChEBI | | Dimethoxon | ChEBI | | O,O-Dimethyl S-((methylcarbamoyl)methyl)phosphorothioate | ChEBI | | O,O-Dimethyl S-(N-methylcarbamoylmethyl) phosphorothiolate | ChEBI | | O,O-Dimethyl S-(N-methylcarbamoylmethyl) thiophosphate | ChEBI | | O,O-Dimethyl S-[(methylcarbamoyl)methyl] phosphorothioate | ChEBI | | O,O-Dimethyl S-[2-(methylamino)-2-oxoethyl] thiophosphate | ChEBI | | O,O-Dimethyl-S-(N-methylcarbamoylmethyl)phosphorothioate | ChEBI | | Phosphorothioic acid, O,O-dimethyl S-(2-(methylamino)-2-oxoethyl) ester | ChEBI | | O,O-Dimethyl S-((methylcarbamoyl)methyl)phosphorothioic acid | Generator | | O,O-Dimethyl S-(N-methylcarbamoylmethyl) phosphorothiolic acid | Generator | | O,O-Dimethyl S-(N-methylcarbamoylmethyl) thiophosphoric acid | Generator | | O,O-Dimethyl S-[(methylcarbamoyl)methyl] phosphorothioic acid | Generator | | O,O-Dimethyl S-[2-(methylamino)-2-oxoethyl] thiophosphoric acid | Generator | | O,O-Dimethyl-S-(N-methylcarbamoylmethyl)phosphorothioic acid | Generator | | Phosphorothioate, O,O-dimethyl S-(2-(methylamino)-2-oxoethyl) ester | Generator | | Omethoic acid | Generator | | 2-Dimethoxyphosphorylsulphanyl-N-methylacetamide | Generator | | O-Methoate | MeSH | | Omethoate | MeSH |
|
|---|
| Chemical Formula | C5H12NO4PS |
|---|
| Average Molecular Mass | 213.190 g/mol |
|---|
| Monoisotopic Mass | 213.022 g/mol |
|---|
| CAS Registry Number | 1113-02-6 |
|---|
| IUPAC Name | 2-[(dimethoxyphosphoryl)sulfanyl]-N-methylethanimidic acid |
|---|
| Traditional Name | omethoate |
|---|
| SMILES | COP(=O)(OC)SCC(O)=NC |
|---|
| InChI Identifier | InChI=1S/C5H12NO4PS/c1-6-5(7)4-12-11(8,9-2)10-3/h4H2,1-3H3,(H,6,7) |
|---|
| InChI Key | PZXOQEXFMJCDPG-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as secondary carboxylic acid amides. Secondary carboxylic acid amides are compounds containing a secondary carboxylic acid amide functional group, with the general structure RC(=O)N(R')H (R,R'=alkyl, aryl). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Carboxylic acid derivatives |
|---|
| Direct Parent | Secondary carboxylic acid amides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Secondary carboxylic acid amide
- Sulfenyl compound
- Organothiophosphorus compound
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-1960000000-32689e71a178f441c46c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-05gi-5910000000-4fe38459de7a4446e793 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00dl-9700000000-ffbdebb0d320224c9bf3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03dl-0950000000-1f0ccdc0728a5ae9fc12 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0292-1910000000-628c2d458a266f7be538 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0002-1900000000-a1f27e057b54908399bd | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Omethoate |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 38730 |
|---|
| PubChem Compound ID | 14210 |
|---|
| Kegg Compound ID | C18662 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|