| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-19 02:25:30 UTC |
|---|
| Update Date | 2016-11-09 01:09:55 UTC |
|---|
| Accession Number | CHEM007428 |
|---|
| Identification |
|---|
| Common Name | ROSIN (PINUS SPP.) AND ROSIN DERIVATIVES |
|---|
| Class | Small Molecule |
|---|
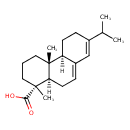
| Description | An abietane diterpenoid that is abieta-7,13-diene substituted by a carboxy group at position 18. |
|---|
| Contaminant Sources | - EAFUS Chemicals
- HPV EPA Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (1R,4AR,10ar)-1,4a-dimethyl-7-(propan-2-yl)-1,2,3,4,4a,5,6,10,10a-decahydrophenanthrene-1-carboxylic acid | ChEBI | | 13-Isopropylpodocarpa-7,13-dien-15-Oic acid | ChEBI | | 7,13-Abietadien-18-Oic acid | ChEBI | | Sylvic acid | ChEBI | | Abieta-7,13-dien-18-Oate | Kegg | | (1R,4AR,10ar)-1,4a-dimethyl-7-(propan-2-yl)-1,2,3,4,4a,5,6,10,10a-decahydrophenanthrene-1-carboxylate | Generator | | 13-Isopropylpodocarpa-7,13-dien-15-Oate | Generator | | 7,13-Abietadien-18-Oate | Generator | | Sylvate | Generator | | Abieta-7,13-dien-18-Oic acid | Generator | | Abietate | Generator | | Abietic acid, 2-aminoethanol | MeSH | | Abietic acid, barium salt | MeSH | | Abietic acid, palladium (+2) salt | MeSH | | Abietic acid, potassium salt | MeSH | | Abietic acid, sodium salt | MeSH | | Abietic acid, strontium salt | MeSH | | Abietic acid, zinc salt | MeSH | | Abietic acid, ammonium salt | MeSH | | Abietic acid, calcium salt | MeSH | | Abietic acid, cobalt salt | MeSH | | Abietic acid, copper salt | MeSH | | (-)-Abietic acid | PhytoBank |
|
|---|
| Chemical Formula | C20H30O2 |
|---|
| Average Molecular Mass | 302.451 g/mol |
|---|
| Monoisotopic Mass | 302.225 g/mol |
|---|
| CAS Registry Number | 8050-09-7 |
|---|
| IUPAC Name | (1R,4aR,4bR,10aR)-1,4a-dimethyl-7-(propan-2-yl)-1,2,3,4,4a,4b,5,6,10,10a-decahydrophenanthrene-1-carboxylic acid |
|---|
| Traditional Name | abietic acid |
|---|
| SMILES | [H][C@]12CCC(=CC1=CC[C@@]1([H])[C@@](C)(CCC[C@]21C)C(O)=O)C(C)C |
|---|
| InChI Identifier | InChI=1S/C20H30O2/c1-13(2)14-6-8-16-15(12-14)7-9-17-19(16,3)10-5-11-20(17,4)18(21)22/h7,12-13,16-17H,5-6,8-11H2,1-4H3,(H,21,22)/t16-,17+,19+,20+/m0/s1 |
|---|
| InChI Key | RSWGJHLUYNHPMX-ONCXSQPRSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as diterpenoids. These are terpene compounds formed by four isoprene units. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Diterpenoids |
|---|
| Direct Parent | Diterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Diterpenoid
- Abietane diterpenoid
- Phenanthrene
- Hydrophenanthrene
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| GC-MS | GC-MS Spectrum - GC-MS (1 TMS) | splash10-052f-4930000000-edf20ffe2ea566e86846 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0197000000-470d0b570779ddd17d57 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4r-2391000000-eda3f35d325884da5dd6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000i-3960000000-247589dc93f8a21aea1c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0049000000-e3e772105a9bb4ad0e1d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0pb9-0094000000-71bdc04831de5f432e38 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052o-1090000000-ddcbe5995de5bd5518d1 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00000871 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Abietic_acid |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 28987 |
|---|
| PubChem Compound ID | 10569 |
|---|
| Kegg Compound ID | C06087 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|