| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-19 02:12:17 UTC |
|---|
| Update Date | 2016-11-09 01:09:42 UTC |
|---|
| Accession Number | CHEM006306 |
|---|
| Identification |
|---|
| Common Name | LIGNIN SODIUM SULFONATE |
|---|
| Class | Small Molecule |
|---|
| Description | |
|---|
| Contaminant Sources | - EAFUS Chemicals
- HPV EPA Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
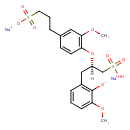
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| SODIUM ligninsulfonic acid | Generator | | SODIUM ligninsulphonate | Generator | | SODIUM ligninsulphonic acid | Generator | | Lignosulfuric acid, sodium salt | MeSH | | Lignosulfonates | MeSH | | Lignosulfuric acid | MeSH | | AHR 2438b | MeSH | | Disodium 2-methoxy-6-[(2R)-2-[2-methoxy-4-(3-sulfonatopropyl)phenoxy]-3-sulfopropyl]benzen-1-olic acid | Generator | | Disodium 2-methoxy-6-[(2R)-2-[2-methoxy-4-(3-sulphonatopropyl)phenoxy]-3-sulphopropyl]benzen-1-olate | Generator | | Disodium 2-methoxy-6-[(2R)-2-[2-methoxy-4-(3-sulphonatopropyl)phenoxy]-3-sulphopropyl]benzen-1-olic acid | Generator |
|
|---|
| Chemical Formula | C20H24Na2O10S2 |
|---|
| Average Molecular Mass | 534.500 g/mol |
|---|
| Monoisotopic Mass | 534.061 g/mol |
|---|
| CAS Registry Number | 8061-51-6 |
|---|
| IUPAC Name | disodium 2-methoxy-6-[(2R)-2-[2-methoxy-4-(3-sulfonatopropyl)phenoxy]-3-sulfopropyl]benzen-1-olate |
|---|
| Traditional Name | disodium 2-methoxy-6-[(2R)-2-[2-methoxy-4-(3-sulfonatopropyl)phenoxy]-3-sulfopropyl]benzenolate |
|---|
| SMILES | [Na+].[Na+].[H][C@@](CC1=C([O-])C(OC)=CC=C1)(CS(O)(=O)=O)OC1=C(OC)C=C(CCCS([O-])(=O)=O)C=C1 |
|---|
| InChI Identifier | InChI=1S/C20H26O10S2.2Na/c1-28-18-7-3-6-15(20(18)21)12-16(13-32(25,26)27)30-17-9-8-14(11-19(17)29-2)5-4-10-31(22,23)24;;/h3,6-9,11,16,21H,4-5,10,12-13H2,1-2H3,(H,22,23,24)(H,25,26,27);;/q;2*+1/p-2/t16-;;/m1../s1 |
|---|
| InChI Key | YDEXUEFDPVHGHE-GGMCWBHBSA-L |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as lignans, neolignans and related compounds. These are plant products of low molecular weight formed primarily from oxidative coupling of two p-propylphenol moieties. They can also be described as micromolecules with two phenylpropanoid units coupled together. They can be attached in various manners, like C5-C5', C8-C8'. Most known natural lignans are oxidized at C9 and C9´ and, based upon the way in which oxygen is incorporated into the skeleton and on the cyclization patterns, a wide range of lignans of very different structural types can be formed. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lignans, neolignans and related compounds |
|---|
| Class | Not Available |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Lignans, neolignans and related compounds |
|---|
| Alternative Parents | |
|---|
| Substituents | - Neolignan skeleton
- Methoxyphenol
- Phenoxy compound
- Methoxybenzene
- Phenol ether
- Anisole
- 1-hydroxy-4-unsubstituted benzenoid
- Phenol
- Alkyl aryl ether
- Benzenoid
- Monocyclic benzene moiety
- Alkanesulfonic acid
- Sulfonyl
- Organosulfonic acid
- Organosulfonic acid or derivatives
- Organic sulfonic acid or derivatives
- Organic alkali metal salt
- Ether
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organic sodium salt
- Organic salt
- Organosulfur compound
- Organooxygen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000b-0010930000-43937a905ebaab1890c0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-003b-0334900000-faae46c9b60badb4739c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00l2-0594400000-08c10e79968d7f6955bf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0000090000-333087a59974e6112155 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001i-0000090000-333087a59974e6112155 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-001i-0000090000-333087a59974e6112155 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 25113562 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|