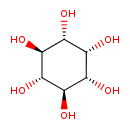
| GC-MS | GC-MS Spectrum - GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (6 TMS) | splash10-014j-0953000000-1571a0577293e96091b1 | Spectrum |
| GC-MS | GC-MS Spectrum - GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (6 TMS) | splash10-00kb-0932000000-96ddc21293431d746ad2 | Spectrum |
| GC-MS | GC-MS Spectrum - GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (Non-derivatized) | splash10-00kb-0942000000-b78076224adf13bade25 | Spectrum |
| GC-MS | GC-MS Spectrum - GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (Non-derivatized) | splash10-00kb-0942000000-42c4e40d04b4f306535d | Spectrum |
| GC-MS | GC-MS Spectrum - GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (6 TMS) | splash10-00di-8942000000-f5d7abd38d6ff547a0a9 | Spectrum |
| GC-MS | GC-MS Spectrum - GC-MS (6 TMS) | splash10-066r-0975000000-df7304c12b6c9bdb0f76 | Spectrum |
| GC-MS | GC-MS Spectrum - GC-EI-TOF (Non-derivatized) | splash10-014j-0953000000-1571a0577293e96091b1 | Spectrum |
| GC-MS | GC-MS Spectrum - GC-EI-TOF (Non-derivatized) | splash10-00kb-0932000000-96ddc21293431d746ad2 | Spectrum |
| GC-MS | GC-MS Spectrum - GC-EI-TOF (Non-derivatized) | splash10-00kb-0942000000-b78076224adf13bade25 | Spectrum |
| GC-MS | GC-MS Spectrum - GC-EI-TOF (Non-derivatized) | splash10-00di-8942000000-f5d7abd38d6ff547a0a9 | Spectrum |
| GC-MS | GC-MS Spectrum - GC-MS (Non-derivatized) | splash10-066r-0975000000-df7304c12b6c9bdb0f76 | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-03dr-9700000000-18a13ab4150e6ebb20cd | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (6 TMS) - 70eV, Positive | splash10-00fr-9001040000-00e67b437c4329aee95d | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_1) - 70eV, Positive | Not Available | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive | Not Available | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_3) - 70eV, Positive | Not Available | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_4) - 70eV, Positive | Not Available | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_5) - 70eV, Positive | Not Available | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_6) - 70eV, Positive | Not Available | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_1) - 70eV, Positive | Not Available | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_2) - 70eV, Positive | Not Available | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_3) - 70eV, Positive | Not Available | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_4) - 70eV, Positive | Not Available | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_5) - 70eV, Positive | Not Available | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, N/A (Annotated) | splash10-0a59-3900000000-96084971c3e13a1e7451 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, N/A (Annotated) | splash10-001i-9000000000-1de17d7f53ca727e6d72 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, N/A (Annotated) | splash10-0zh9-9000000000-bef910214341056a78aa | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Orbitrap 2V, negative | splash10-004i-0900000000-41a49af846b1636763b5 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Orbitrap 2V, negative | splash10-004i-0900000000-06f9ecfccf30d40ef204 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Orbitrap 3V, negative | splash10-004i-0900000000-bca88f6782a8ed386223 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Orbitrap 4V, negative | splash10-004i-1900000000-500ab195d69568b8678b | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Orbitrap 5V, negative | splash10-01ti-5900000000-a1657b54c985e2be1d19 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Orbitrap 6V, negative | splash10-000i-9600000000-325cbb8dbfdeafb2b31f | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Orbitrap 8V, negative | splash10-000i-9200000000-9145a4766351204e723a | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Orbitrap 9V, negative | splash10-000i-9100000000-2bbbcfa4733b749ec768 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Orbitrap 10V, negative | splash10-000i-9000000000-de0ec8731a152b612bb9 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Orbitrap 12V, negative | splash10-0079-9000000000-41d049d1feef94e190d4 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Orbitrap 14V, negative | splash10-059i-9000000000-796e728daf28d5122dee | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Orbitrap 15V, negative | splash10-059i-9000000000-8643e206319f3008a8d4 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Orbitrap 18V, negative | splash10-0abi-9000000000-98e61f19bc2842f2e97b | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - Orbitrap 20V, negative | splash10-0ab9-9000000000-29aa062fc4d5341167b6 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - n/a 12V, negative | splash10-03di-1900000000-5a2897fd032a959918b3 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - n/a 12V, negative | splash10-000i-9000000000-71c4786b7034d5a0a0a9 | Spectrum |
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0900000000-5cb39f92646251665ebf | Spectrum |
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0900000000-2aeb7708f31c772e2d95 | Spectrum |
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-08gi-8900000000-74f89711a1c6891808a4 | Spectrum |
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0900000000-8b68ff47f846a8ca1ddd | Spectrum |
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-1900000000-03ef8572289795275d9c | Spectrum |
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0570-9300000000-950a37bd331028801e9f | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 2D NMR | [1H,1H] 2D NMR Spectrum | Not Available | Spectrum |
| 2D NMR | [1H,13C] 2D NMR Spectrum | Not Available | Spectrum |