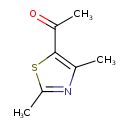| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-19 02:01:42 UTC |
|---|
| Update Date | 2016-11-09 01:09:32 UTC |
|---|
| Accession Number | CHEM005395 |
|---|
| Identification |
|---|
| Common Name | 2,4-DIMETHYL-5-ACETYLTHIAZOLE |
|---|
| Class | Small Molecule |
|---|
| Description | 5-Acetyl-2,4-dimethylthiazole is a flavouring ingredient. |
|---|
| Contaminant Sources | - EAFUS Chemicals
- FooDB Chemicals
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1-(2,4-Dimethyl-1,3-thiazol-5-yl)ethanone | HMDB | | 1-(2,4-Dimethyl-5-thiazolyl)-ethanone | HMDB | | 1-(2,4-Dimethyl-5-thiazolyl)ethanone | HMDB | | 1-(2,4-Dimethyl-5-thiazolyl)ethanone, 9ci | HMDB | | 1-(2,4-Dimethylthiazol-5-yl)ethan-1-one | HMDB | | 2,4-Dimethyl-5-acetylthiazole | HMDB | | 2,4-Dimethyl-5-thiazolyl methyl ketone | HMDB | | 2,4-Dimethyl-5-thiazoyl methyl ketone | HMDB | | 5-Acetyl-2,4-dimethyltriazole | HMDB | | FEMA 3267 | HMDB | | Ketone, 2,4-dimethyl-5-thiazolyl methyl | HMDB | | Thiazole, 5-acetyl-2,4-dimethyl | HMDB |
|
|---|
| Chemical Formula | C7H9NOS |
|---|
| Average Molecular Mass | 155.217 g/mol |
|---|
| Monoisotopic Mass | 155.040 g/mol |
|---|
| CAS Registry Number | 38205-60-6 |
|---|
| IUPAC Name | 1-(dimethyl-1,3-thiazol-5-yl)ethan-1-one |
|---|
| Traditional Name | 1-(dimethyl-1,3-thiazol-5-yl)ethanone |
|---|
| SMILES | CC(=O)C1=C(C)N=C(C)S1 |
|---|
| InChI Identifier | InChI=1S/C7H9NOS/c1-4-7(5(2)9)10-6(3)8-4/h1-3H3 |
|---|
| InChI Key | BLQOKWQUTLNKON-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 2,4,5-trisubstituted thiazoles. 2,4,5-trisubstituted thiazoles are compounds containing a thiazole ring substituted at positions 2, 4 and 5 only. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Azoles |
|---|
| Sub Class | Thiazoles |
|---|
| Direct Parent | 2,4,5-trisubstituted thiazoles |
|---|
| Alternative Parents | |
|---|
| Substituents | - Aryl alkyl ketone
- Aryl ketone
- 2,4,5-trisubstituted 1,3-thiazole
- Heteroaromatic compound
- Ketone
- Azacycle
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0006-8900000000-b5081d4c33feb5749a6e | Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0006-8900000000-b5081d4c33feb5749a6e | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-08fu-6900000000-7446bb364608a5a41b2d | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4r-0900000000-ec5707a16e4511841424 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0900000000-43f9721bf782523cf986 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00kr-6900000000-1e67714000444db6d827 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0900000000-00bd0ffce00b7b3e8e65 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0900000000-437cb2efc4e93a994c1b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-03dl-9400000000-95f049187db571181d05 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0900000000-e187059cc071e9e4229c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4l-9800000000-8333fbc21f9e6d60a300 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-006x-9100000000-6d9549978c039209807b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0900000000-ed1ef2ec5c8c2dcf3778 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0ik9-5900000000-c8ca822b600ce6908fa9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-022c-9400000000-89d52c6a2eac34c50011 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0041484 |
|---|
| FooDB ID | FDB021449 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 454346 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 520888 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|