| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-19 01:54:48 UTC |
|---|
| Update Date | 2016-10-28 10:00:56 UTC |
|---|
| Accession Number | CHEM004785 |
|---|
| Identification |
|---|
| Common Name | ASPERGILLUS NIGER FOR FERMENTATION PRODUCTION OF CITRIC ACID |
|---|
| Class | Small Molecule |
|---|
| Description | The simplest monocarboxylic acid amide, obtained by formal condensation of formic acid with ammonia. The parent of the class of formaldehydes. |
|---|
| Contaminant Sources | - DEA Chemicals
- EAFUS Chemicals
- FooDB Chemicals
- HMDB Contaminants - Feces
- HPV EPA Chemicals
- OECD HPV Chemicals
- STOFF IDENT Compounds
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
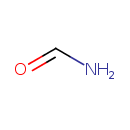
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Ameisensaeureamid | ChEBI | | Carbamaldehyde | ChEBI | | Formamid | ChEBI | | Formimidic acid | ChEBI | | Methanamid | ChEBI | | Methanamide | ChEBI | | Formimidate | Generator |
|
|---|
| Chemical Formula | CH3NO |
|---|
| Average Molecular Mass | 45.041 g/mol |
|---|
| Monoisotopic Mass | 45.021 g/mol |
|---|
| CAS Registry Number | 9001-23-4 |
|---|
| IUPAC Name | formamide |
|---|
| Traditional Name | formamide |
|---|
| SMILES | NC=O |
|---|
| InChI Identifier | InChI=1S/CH3NO/c2-1-3/h1H,(H2,2,3) |
|---|
| InChI Key | ZHNUHDYFZUAESO-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as carboximidic acids. These are organic acids with the general formula RC(=N)-OH (R=H, organic group). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboximidic acids and derivatives |
|---|
| Sub Class | Carboximidic acids |
|---|
| Direct Parent | Carboximidic acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Carboximidic acid
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Imine
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0002-9000000000-f6326cc6c67041284b52 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-9000000000-4a58cc52ed1f85a41b4c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-9000000000-52b8e3cce362934e1d52 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004j-9000000000-32c53cf62ffc73e10c84 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-9000000000-0a22a0222cfe33823b8b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-9000000000-2473e800b4396749f887 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004l-9000000000-3221c668103b22c85e42 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-9000000000-e0e93b80e95c0e39a985 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-9000000000-e0e93b80e95c0e39a985 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0002-9000000000-a08824ab04647a23f4ad | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-9000000000-451c209790660c80803f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-9000000000-451c209790660c80803f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9000000000-ab072a75b4f755c7548c | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 2D NMR | [1H,1H] 2D NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0001536 |
|---|
| FooDB ID | FDB022677 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00011920 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | FORMAMIDE |
|---|
| METLIN ID | 4182 |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Formamide |
|---|
| Chemspider ID | 693 |
|---|
| ChEBI ID | 16397 |
|---|
| PubChem Compound ID | 713 |
|---|
| Kegg Compound ID | C00488 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | ECMDB01536 |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. https://www.ncbi.nlm.nih.gov/pubmed/?term=11282235 | | 2. https://www.ncbi.nlm.nih.gov/pubmed/?term=11545392 | | 3. https://www.ncbi.nlm.nih.gov/pubmed/?term=12115814 | | 4. https://www.ncbi.nlm.nih.gov/pubmed/?term=14750843 | | 5. https://www.ncbi.nlm.nih.gov/pubmed/?term=15082074 | | 6. https://www.ncbi.nlm.nih.gov/pubmed/?term=17184725 | | 7. https://www.ncbi.nlm.nih.gov/pubmed/?term=19334838 | | 8. https://www.ncbi.nlm.nih.gov/pubmed/?term=21215846 | | 9. https://www.ncbi.nlm.nih.gov/pubmed/?term=21229996 | | 10. https://www.ncbi.nlm.nih.gov/pubmed/?term=21573300 | | 11. https://www.ncbi.nlm.nih.gov/pubmed/?term=21647491 | | 12. https://www.ncbi.nlm.nih.gov/pubmed/?term=21647492 | | 13. https://www.ncbi.nlm.nih.gov/pubmed/?term=21769603 | | 14. https://www.ncbi.nlm.nih.gov/pubmed/?term=21932847 | | 15. Magill, P. L. Formamide. Journal of Industrial and Engineering Chemistry (Washington, D. C.) (1934), 26 611-14. | | 16. Magill, P. L. Formamide. Journal of Industrial and Engineering Chemistry (Washington, D. C.) (1934), 26 611-14. | | 17. Lareo AC, Perbellini L: Biological monitoring of workers exposed to N-N-dimethylformamide. II. Dimethylformamide and its metabolites in urine of exposed workers. Int Arch Occup Environ Health. 1995;67(1):47-52. | | 18. Kawashima K, Doi H, Ito Y, Shibata MA, Yoshinaka R, Otsuki Y: Evaluation of cell death and proliferation in psoriatic epidermis. J Dermatol Sci. 2004 Sep;35(3):207-14. | | 19. Major J, Hudak A, Kiss G, Jakab MG, Szaniszlo J, Naray M, Nagy I, Tompa A: Follow-up biological and genotoxicological monitoring of acrylonitrile- and dimethylformamide-exposed viscose rayon plant workers. Environ Mol Mutagen. 1998;31(4):301-10. | | 20. Al-Soud WA, Ouis IS, Li DQ, Ljungh S, Wadstrom T: Characterization of the PCR inhibitory effect of bile to optimize real-time PCR detection of Helicobacter species. FEMS Immunol Med Microbiol. 2005 May 1;44(2):177-82. | | 21. Eizuru Y, Minamishima Y, Matsumoto T, Hamakado T, Mizukoshi M, Nabeshima K, Koono M, Yoshida A, Yoshida H, Kikuchi M: Application of in situ hybridization with a novel phenytoin-labeled probe to conventional formalin-fixed, paraffin-embedded tissue sections. J Virol Methods. 1995 Apr;52(3):309-16. | | 22. Walrath J, Fayerweather WE, Gilby PG, Pell S: A case-control study of cancer among du pont employees with potential for exposure to dimethylformamide. J Occup Med. 1989 May;31(5):432-8. |
|
|---|