| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-19 01:54:40 UTC |
|---|
| Update Date | 2016-11-09 01:09:25 UTC |
|---|
| Accession Number | CHEM004770 |
|---|
| Identification |
|---|
| Common Name | L-ARABINOSE |
|---|
| Class | Small Molecule |
|---|
| Description | L-Arabinose, also known as L-arabinopyranose, belongs to the class of organic compounds known as pentoses. These are monosaccharides in which the carbohydrate moiety contains five carbon atoms. L-Arabinose exists as a solid, possibly soluble (in water), and an extremely weak basic (essentially neutral) compound (based on its pKa) molecule. L-Arabinose exists in all living species, ranging from bacteria to humans. L-Arabinose has been found to be associated with several diseases known as eosinophilic esophagitis and colorectal cancer; also l-arabinose has been linked to the inborn metabolic disorders including ribose-5-phosphate isomerase deficiency. |
|---|
| Contaminant Sources | - EAFUS Chemicals
- FooDB Chemicals
- HMDB Contaminants - Urine
|
|---|
| Contaminant Type | Not Available |
|---|
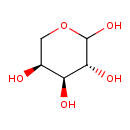
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| L-Arabinopyranose | Kegg | | Arabinose | HMDB | | L-(+)-Arabinose | HMDB | | L Arabinose | HMDB | | Pectinose | PhytoBank |
|
|---|
| Chemical Formula | C5H10O5 |
|---|
| Average Molecular Mass | 150.130 g/mol |
|---|
| Monoisotopic Mass | 150.053 g/mol |
|---|
| CAS Registry Number | 5328-37-0 |
|---|
| IUPAC Name | (3R,4S,5S)-oxane-2,3,4,5-tetrol |
|---|
| Traditional Name | L-arabinopyranose |
|---|
| SMILES | OC[C@H](O)[C@H](O)[C@@H](O)C=O |
|---|
| InChI Identifier | InChI=1S/C5H10O5/c6-1-3(8)5(10)4(9)2-7/h1,3-5,7-10H,2H2/t3-,4-,5+/m0/s1 |
|---|
| InChI Key | PYMYPHUHKUWMLA-VAYJURFESA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as pentoses. These are monosaccharides in which the carbohydrate moiety contains five carbon atoms. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Pentoses |
|---|
| Alternative Parents | |
|---|
| Substituents | - Pentose monosaccharide
- Oxane
- Secondary alcohol
- Hemiacetal
- Oxacycle
- Organoheterocyclic compound
- Polyol
- Hydrocarbon derivative
- Alcohol
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-05al-9300000000-73d0d716ea47fc77a7ae | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (4 TMS) - 70eV, Positive | splash10-00g0-9256300000-ae2d57025e3828494249 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0ue9-1900000000-07783f4be8db8a14f6f0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0f89-1900000000-510acee1186ebf4c526b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-07s1-9100000000-ff1e6f260ccdb7260ded | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-2900000000-f624316a77fb9d1bc74f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001j-4900000000-768143fc08314c41644f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9000000000-13182c55ee979014fcfa | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 2D NMR | [1H,13C] 2D NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0000646 |
|---|
| FooDB ID | FDB012306 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00041349 |
|---|
| BiGG ID | 34429 |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | 5474 |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Arabinose |
|---|
| Chemspider ID | 388335 |
|---|
| ChEBI ID | 17535 |
|---|
| PubChem Compound ID | 439195 |
|---|
| Kegg Compound ID | C00259 |
|---|
| YMDB ID | YMDB00893 |
|---|
| ECMDB ID | ECMDB00646 |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. Whistler, Roy L.; Schweiger, Richard. Preparation of D-arabinose from D-glucose with hypochlorite. Journal of the American Chemical Society (1959), 81 5190-2. | | 2. GARTON GA: THE COMPOSITION AND BIOSYNTHESIS OF MILK LIPIDS. J Lipid Res. 1963 Jul;4:237-54. | | 3. Qian L, Zhao A, Zhang Y, Chen T, Zeisel SH, Jia W, Cai W: Metabolomic Approaches to Explore Chemical Diversity of Human Breast-Milk, Formula Milk and Bovine Milk. Int J Mol Sci. 2016 Dec 17;17(12). pii: ijms17122128. doi: 10.3390/ijms17122128. | | 4. Whistler, Roy L.; Schweiger, Richard. Preparation of D-arabinose from D-glucose with hypochlorite. Journal of the American Chemical Society (1959), 81 5190-2. | | 5. Lobley RW, Burrows PC, Warwick R, Dawson DJ, Holmes R: Simultaneous assessment of intestinal permeability and lactose tolerance with orally administered raffinose, lactose and L-arabinose. Clin Sci (Lond). 1990 Aug;79(2):175-83. | | 6. Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, Omenn GS, Ghosh D, Pennathur S, Alexander DC, Berger A, Shuster JR, Wei JT, Varambally S, Beecher C, Chinnaiyan AM: Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009 Feb 12;457(7231):910-4. doi: 10.1038/nature07762. | | 7. Osaki S, Kimura T, Sugimoto T, Hizukuri S, Iritani N: L-arabinose feeding prevents increases due to dietary sucrose in lipogenic enzymes and triacylglycerol levels in rats. J Nutr. 2001 Mar;131(3):796-9. | | 8. Seri K, Sanai K, Matsuo N, Kawakubo K, Xue C, Inoue S: L-arabinose selectively inhibits intestinal sucrase in an uncompetitive manner and suppresses glycemic response after sucrose ingestion in animals. Metabolism. 1996 Nov;45(11):1368-74. | | 9. Schutte JB, de Jong J, van Weerden EJ, Tamminga S: Nutritional implications of L-arabinose in pigs. Br J Nutr. 1992 Jul;68(1):195-207. | | 10. Shaw W, Kassen E, Chaves E: Increased urinary excretion of analogs of Krebs cycle metabolites and arabinose in two brothers with autistic features. Clin Chem. 1995 Aug;41(8 Pt 1):1094-104. | | 11. TOUSTER O, HARWELL SO: The isolation of L-arabitol from pentosuric urine. J Biol Chem. 1958 Feb;230(2):1031-41. | | 12. Onkenhout W, Groener JE, Verhoeven NM, Yin C, Laan LA: L-Arabinosuria: a new defect in human pentose metabolism. Mol Genet Metab. 2002 Sep-Oct;77(1-2):80-5. | | 13. Ahmed Z, Shimonishi T, Bhuiyan SH, Utamura M, Takada G, Izumori K: Biochemical preparation of L-ribose and L-arabinose from ribitol: a new approach. J Biosci Bioeng. 1999;88(4):444-8. | | 14. https://www.ncbi.nlm.nih.gov/pubmed/?term=1177062 | | 15. https://www.ncbi.nlm.nih.gov/pubmed/?term=17336832 | | 16. https://www.ncbi.nlm.nih.gov/pubmed/?term=19913595 | | 17. https://www.ncbi.nlm.nih.gov/pubmed/?term=28785254 |
|
|---|