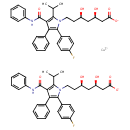
| Atorvastan | ChEBI |
| Calcium (betar,deltar)-2-(p-fluorophenyl)-beta,delta-dihydroxy-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)pyrrole-1-heptanoate (1:2) | ChEBI |
| Lipitor | ChEBI |
| Liprimar | ChEBI |
| Calcium (betar,deltar)-2-(p-fluorophenyl)-b,delta-dihydroxy-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)pyrrole-1-heptanoate (1:2) | Generator |
| Calcium (betar,deltar)-2-(p-fluorophenyl)-b,delta-dihydroxy-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)pyrrole-1-heptanoic acid (1:2) | Generator |
| Calcium (betar,deltar)-2-(p-fluorophenyl)-beta,delta-dihydroxy-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)pyrrole-1-heptanoic acid (1:2) | Generator |
| Calcium (betar,deltar)-2-(p-fluorophenyl)-β,δ-dihydroxy-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)pyrrole-1-heptanoate (1:2) | Generator |
| Calcium (betar,deltar)-2-(p-fluorophenyl)-β,δ-dihydroxy-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)pyrrole-1-heptanoic acid (1:2) | Generator |
| Calcium (betar,deltar)-2-(P-fluorophenyl)-b,δ-dihydroxy-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)pyrrole-1-heptanoate (1:2) | Generator |
| Calcium (betar,deltar)-2-(P-fluorophenyl)-b,δ-dihydroxy-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)pyrrole-1-heptanoic acid (1:2) | Generator |
| Calcium, atorvastatin | MeSH |
| Trihydrate, atorvastatin calcium | MeSH |
| Atorvastatin calcium trihydrate | MeSH |
| Liptonorm | MeSH |
| Calcium salt atorvastatin | MeSH |
| Anhydrous, atorvastatin calcium | MeSH |
| Calcium hydrate, atorvastatin | MeSH |
| Calcium trihydrate, atorvastatin | MeSH |
| Hydrate, atorvastatin calcium | MeSH |
| Atorvastatin calcium anhydrous | MeSH |
| Atorvastatin, calcium salt | MeSH |
| Calcium anhydrous, atorvastatin | MeSH |
| Atorvastatin | MeSH |
| Atorvastatin calcium hydrate | MeSH |
| (3R,5R)-7-(2-(4-Fluorophenyl)-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)-1H-pyrrol-1-yl)-3,5-dihydroxyheptanoic acid | MeSH |