| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-19 01:36:18 UTC |
|---|
| Update Date | 2016-11-09 01:09:17 UTC |
|---|
| Accession Number | CHEM004163 |
|---|
| Identification |
|---|
| Common Name | Fensulfothion |
|---|
| Class | Small Molecule |
|---|
| Description | |
|---|
| Contaminant Sources | - Clean Air Act Chemicals
- My Exposome Chemicals
- STOFF IDENT Compounds
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
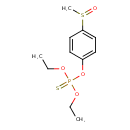
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Dasanit | ChEBI | | O,O-Diethyl O-(p-(methylsulfinyl)phenyl) phosphorothioate | ChEBI | | O,O-Diethyl O-4-methylsulphinylphenyl phosphorothioate | ChEBI | | O,O-Diethyl O-[4-(methylsulfinyl)phenyl] thiophosphate | ChEBI | | O,O-Diethyl O-p-(methylsulfinyl)phenyl thiophosphate | ChEBI | | Phosphorothioic acid, O,O-diethyl O-(4-(methylsulfinyl)phenyl) ester | ChEBI | | O,O-Diethyl O-(p-(methylsulfinyl)phenyl) phosphorothioic acid | Generator | | O,O-Diethyl O-(p-(methylsulphinyl)phenyl) phosphorothioate | Generator | | O,O-Diethyl O-(p-(methylsulphinyl)phenyl) phosphorothioic acid | Generator | | O,O-Diethyl O-4-methylsulfinylphenyl phosphorothioate | Generator | | O,O-Diethyl O-4-methylsulfinylphenyl phosphorothioic acid | Generator | | O,O-Diethyl O-4-methylsulphinylphenyl phosphorothioic acid | Generator | | O,O-Diethyl O-[4-(methylsulfinyl)phenyl] thiophosphoric acid | Generator | | O,O-Diethyl O-[4-(methylsulphinyl)phenyl] thiophosphate | Generator | | O,O-Diethyl O-[4-(methylsulphinyl)phenyl] thiophosphoric acid | Generator | | O,O-Diethyl O-p-(methylsulfinyl)phenyl thiophosphoric acid | Generator | | O,O-Diethyl O-p-(methylsulphinyl)phenyl thiophosphate | Generator | | O,O-Diethyl O-p-(methylsulphinyl)phenyl thiophosphoric acid | Generator | | Phosphorothioate, O,O-diethyl O-(4-(methylsulfinyl)phenyl) ester | Generator | | Phosphorothioate, O,O-diethyl O-(4-(methylsulphinyl)phenyl) ester | Generator | | Phosphorothioic acid, O,O-diethyl O-(4-(methylsulphinyl)phenyl) ester | Generator | | Fensulphothion | Generator | | Diethoxy-(4-methylsulphinylphenoxy)-sulphanylidene-$l^{5}-phosphane | Generator | | Dansit | MeSH | | Fensulfothion | MeSH |
|
|---|
| Chemical Formula | C11H17O4PS2 |
|---|
| Average Molecular Mass | 308.350 g/mol |
|---|
| Monoisotopic Mass | 308.031 g/mol |
|---|
| CAS Registry Number | 115-90-2 |
|---|
| IUPAC Name | O,O-diethyl O-4-methanesulfinylphenyl phosphorothioate |
|---|
| Traditional Name | fensulfothion |
|---|
| SMILES | CCOP(=S)(OCC)OC1=CC=C(C=C1)S(C)=O |
|---|
| InChI Identifier | InChI=1S/C11H17O4PS2/c1-4-13-16(17,14-5-2)15-10-6-8-11(9-7-10)18(3)12/h6-9H,4-5H2,1-3H3 |
|---|
| InChI Key | XDNBJTQLKCIJBV-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenyl thiophosphates. These are organothiophosphorus compounds that contain a thiophosphoric acid O-esterified with a phenyl group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Organic thiophosphoric acids and derivatives |
|---|
| Sub Class | Thiophosphoric acid esters |
|---|
| Direct Parent | Phenyl thiophosphates |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenyl thiophosphate
- Phenyl sulfoxide
- Phenoxy compound
- Thiophosphate triester
- Benzenoid
- Monocyclic benzene moiety
- Sulfoxide
- Sulfinyl compound
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organosulfur compound
- Organooxygen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a59-1197000000-9c35fc5670964a25ed8b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0zgi-1390000000-fe98f3a3118cbb9136a7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00tu-4940000000-db4f5fda7cf13969392e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0bvi-2294000000-4b8fce8ca0add7c0bdd5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0h00-1290000000-18208837a7c89e7b22b1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0udr-3690000000-bac4ee6bcde4df2da26b | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Fensulfothion |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 34760 |
|---|
| PubChem Compound ID | 8292 |
|---|
| Kegg Compound ID | C14510 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|