| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-19 01:36:09 UTC |
|---|
| Update Date | 2016-11-09 01:09:17 UTC |
|---|
| Accession Number | CHEM004158 |
|---|
| Identification |
|---|
| Common Name | Azaserine |
|---|
| Class | Small Molecule |
|---|
| Description | A carboxylic ester resulting from the formal condensation of the carboxy group of diazoacetic acid with the alcoholic hydroxy group of L-serine. An antibiotic produced by a Streptomyces species. |
|---|
| Contaminant Sources | - Clean Air Act Chemicals
- IARC Carcinogens Group 2B
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Azaserina | ChEBI | | Azaserinum | ChEBI | | L-Azaserine | ChEBI | | L-beta-(Diazoacetoxy)alanin | ChEBI | | L-beta-(Diazoacetoxy)alanine | ChEBI | | L-Serine diazoacetate | ChEBI | | L-Serine diazoacetate ester | ChEBI | | L-b-(Diazoacetoxy)alanin | Generator | | L-Β-(diazoacetoxy)alanin | Generator | | L-b-(Diazoacetoxy)alanine | Generator | | L-Β-(diazoacetoxy)alanine | Generator | | L-Serine diazoacetic acid | Generator | | L-Serine diazoacetic acid ester | Generator | | Azeserine | MeSH | | O Diazoacetyl L serine | MeSH | | O-Diazoacetyl-L-serine | MeSH |
|
|---|
| Chemical Formula | C5H7N3O4 |
|---|
| Average Molecular Mass | 173.127 g/mol |
|---|
| Monoisotopic Mass | 173.044 g/mol |
|---|
| CAS Registry Number | 115-02-6 |
|---|
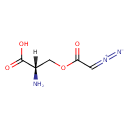
| IUPAC Name | (2S)-2-amino-3-[(2-diazoacetyl)oxy]propanoic acid |
|---|
| Traditional Name | azaserine |
|---|
| SMILES | [H][C@](N)(COC(=O)C=[N+]=[N-])C(O)=O |
|---|
| InChI Identifier | InChI=1S/C5H7N3O4/c6-3(5(10)11)2-12-4(9)1-8-7/h1,3H,2,6H2,(H,10,11)/t3-/m0/s1 |
|---|
| InChI Key | MZZGOOYMKKIOOX-VKHMYHEASA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as l-alpha-amino acids. These are alpha amino acids which have the L-configuration of the alpha-carbon atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | L-alpha-amino acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - L-alpha-amino acid
- Alpha-diazo ester
- Dicarboxylic acid or derivatives
- Carboxylic acid ester
- Amino acid
- Diazo compound
- Carboxylic acid
- Hydrocarbon derivative
- Organic zwitterion
- Organic nitrogen compound
- Primary amine
- Organooxygen compound
- Organonitrogen compound
- Primary aliphatic amine
- Organopnictogen compound
- Organic oxide
- Carbonyl group
- Amine
- Organic oxygen compound
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-0900000000-10846d1a7d038a8128e6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01c9-2900000000-02c0e660d40ae28a1b37 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9100000000-fc49726cce3824e177bf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0900000000-c8afbc846eb1423d47ed | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-4900000000-0e9657b778bfb9ec1b86 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00di-9000000000-861615d60cd01133d065 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | L-AZASERINE |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Azaserine |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 74846 |
|---|
| PubChem Compound ID | 460129 |
|---|
| Kegg Compound ID | C19194 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|