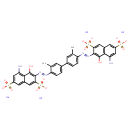
Identification Common Name Trypan blue Class Small Molecule Description An organosulfonate salt that is the tetrasodium salt of 3,3'-bis(5-amino-4-hydroxynaphthalene-2,7-disulfonic acid). Contaminant Sources Clean Air Act Chemicals
HPV EPA Chemicals
IARC Carcinogens Group 2B
ToxCast & Tox21 Chemicals Contaminant Type Not Available Chemical Structure Synonyms Value Source C.I. 23850 ChEBI C.I. direct blue 14 ChEBI C.I. direct blue 14, tetrasodium salt ChEBI Direct blue 14 ChEBI Niagara blue 3b ChEBI Sodium ditolyl-diazobis-8-amino-1-naphthol-3,6-disulfonate ChEBI Sodium ditolyldisazobis-8-amino-1-naphthol-3,6-disulfonate ChEBI Sodium ditolyldisazobis-8-amino-1-naphthol-3,6-disulphonate ChEBI Sodium ditolyl-diazobis-8-amino-1-naphthol-3,6-disulfonic acid Generator Sodium ditolyl-diazobis-8-amino-1-naphthol-3,6-disulphonate Generator Sodium ditolyl-diazobis-8-amino-1-naphthol-3,6-disulphonic acid Generator Sodium ditolyldisazobis-8-amino-1-naphthol-3,6-disulfonic acid Generator Sodium ditolyldisazobis-8-amino-1-naphthol-3,6-disulphonic acid Generator
Chemical Formula C34 H24 N6 Na4 O14 S4 Average Molecular Mass 960.790 g/mol Monoisotopic Mass 959.982 g/mol CAS Registry Number 72-57-1 IUPAC Name tetrasodium 5-amino-3-[(E)-2-{4'-[(E)-2-(8-amino-1-hydroxy-3,6-disulfonatonaphthalen-2-yl)diazen-1-yl]-3,3'-dimethyl-[1,1'-biphenyl]-4-yl}diazen-1-yl]-4-hydroxynaphthalene-2,7-disulfonate Traditional Name tetrasodium 5-amino-3-[(E)-2-{4'-[(E)-2-(8-amino-1-hydroxy-3,6-disulfonatonaphthalen-2-yl)diazen-1-yl]-3,3'-dimethyl-[1,1'-biphenyl]-4-yl}diazen-1-yl]-4-hydroxynaphthalene-2,7-disulfonate SMILES [Na+].[Na+].[Na+].[Na+].CC1=CC(=CC=C1\N=N\C1=C(O)C2=C(N)C=C(C=C2C=C1S([O-])(=O)=O)S([O-])(=O)=O)C1=CC=C(\N=N\C2=C(O)C3=C(C=C(C=C3N)S([O-])(=O)=O)C=C2S([O-])(=O)=O)C(C)=C1 InChI Identifier InChI=1S/C34H28N6O14S4.4Na/c1-15-7-17(3-5-25(15)37-39-31-27(57(49,50)51)11-19-9-21(55(43,44)45)13-23(35)29(19)33(31)41)18-4-6-26(16(2)8-18)38-40-32-28(58(52,53)54)12-20-10-22(56(46,47)48)14-24(36)30(20)34(32)42;;;;/h3-14,41-42H,35-36H2,1-2H3,(H,43,44,45)(H,46,47,48)(H,49,50,51)(H,52,53,54);;;;/q;4*+1/p-4/b39-37+,40-38+;;;; InChI Key GLNADSQYFUSGOU-GPTZEZBUSA-J Chemical Taxonomy Description belongs to the class of organic compounds known as 3,3'-disubstituted benzidines. These are organic compounds containing a benzidine skeleton, which is substituted only at the 3- and 3'-positions. Kingdom Organic compounds Super Class Benzenoids Class Benzene and substituted derivatives Sub Class Biphenyls and derivatives Direct Parent 3,3'-disubstituted benzidines Alternative Parents Substituents 3,3'-disubstituted benzidine
Naphthalene sulfonate
2-naphthalene sulfonic acid or derivatives
Naphthalene sulfonic acid or derivatives
2-naphthalene sulfonate
1-naphthol
Naphthalene
Arylsulfonic acid or derivatives
1-sulfo,2-unsubstituted aromatic compound
1-hydroxy-4-unsubstituted benzenoid
Toluene
Organic sulfonic acid or derivatives
Organosulfonic acid or derivatives
Organosulfonic acid
Sulfonyl
Azo compound
Propargyl-type 1,3-dipolar organic compound
Organic 1,3-dipolar compound
Organic alkali metal salt
Hydrocarbon derivative
Organic salt
Organic sodium salt
Primary amine
Organosulfur compound
Organooxygen compound
Organonitrogen compound
Organic nitrogen compound
Organic oxide
Organopnictogen compound
Organic oxygen compound
Amine
Aromatic homopolycyclic compound Molecular Framework Aromatic homopolycyclic compounds External Descriptors