Identification Common Name Forskolin Class Small Molecule Description Potent activator of the adenylate cyclase system and the biosynthesis of cyclic AMP. From the plant Coleus forskohlii. Has antihypertensive, positive ionotropic, platelet aggregation inhibitory, and smooth muscle relaxant activities; also lowers intraocular pressure and promotes release of hormones from the pituitary gland. Contaminant Sources T3DB toxins
ToxCast & Tox21 Chemicals Contaminant Type Ester
Ether
Natural Compound
Organic Compound
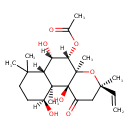
Plant Toxin Chemical Structure Synonyms Value Source 7beta-Acetoxy-8,13-epoxy-1alpha,6beta,9alpha-trihydroxylabd-14-en-11-one ChEBI Coleonol ChEBI Coleonolk ChEBI Colforsina ChEBI Colforsine ChEBI Colforsinum ChEBI Forskolin Kegg 7b-Acetoxy-8,13-epoxy-1a,6b,9a-trihydroxylabd-14-en-11-one Generator 7Β-acetoxy-8,13-epoxy-1α,6β,9α-trihydroxylabd-14-en-11-one Generator Colforsin ChEBI N,N-Dimethyl-beta-alanine-5-(acetyloxy)-3-ethenyldodecahydro-10,10b-dihydroxy-3,4a,7,7,10a-pentamethyl-1-oxo-1H-naphtho(2,1-b)pyran-6-yl ester HCL MeSH NKH 477 MeSH NKH-477 MeSH
Chemical Formula C22 H34 O7 Average Molecular Mass 410.501 g/mol Monoisotopic Mass 410.230 g/mol CAS Registry Number 64657-11-0 and 66575-29-9 IUPAC Name (3R,4aR,5S,6S,6aS,10S,10aR,10bS)-3-ethenyl-6,10,10b-trihydroxy-3,4a,7,7,10a-pentamethyl-1-oxo-dodecahydro-1H-naphtho[2,1-b]pyran-5-yl acetate Traditional Name forskolin SMILES [H][C@]1(O)CCC(C)(C)[C@]2([H])[C@]([H])(O)[C@]([H])(OC(C)=O)[C@@]3(C)O[C@](C)(CC(=O)[C@]3(O)[C@@]12C)C=C InChI Identifier InChI=1S/C22H34O7/c1-8-19(5)11-14(25)22(27)20(6)13(24)9-10-18(3,4)16(20)15(26)17(28-12(2)23)21(22,7)29-19/h8,13,15-17,24,26-27H,1,9-11H2,2-7H3/t13-,15-,16-,17-,19-,20-,21+,22-/m0/s1 InChI Key OHCQJHSOBUTRHG-KGGHGJDLSA-N