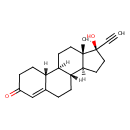
| (17alpha)-17-Ethynyl-17-hydroxyestra-4,8(14),9-trien-3-one | ChEBI |
| 17-alpha-Ethynyl-17-hydroxy-4-estren-3-one | ChEBI |
| 17-alpha-Ethynyl-19-norandrost-4-en-17-beta-ol-3-one | ChEBI |
| 17-alpha-Ethynyl-19-nortestosterone | ChEBI |
| 17-alpha-Ethynyl-4-estren-17-ol-3-one | ChEBI |
| 17-beta-Hydroxy-19-norpregn-4-en-20-yn-3-one | ChEBI |
| 17-Hydroxy-19-nor-17-alpha-pregn-4-en-20-yn-3-one | ChEBI |
| 17-Hydroxy-19-nor-17alpha-pregn-4-en-20-yn-3-one | ChEBI |
| 17alpha-Ethinyl-19-nortestosterone | ChEBI |
| 17alpha-Ethinylestra-4-en-17beta-ol-3-one | ChEBI |
| 17alpha-Ethynyl-17-hydroxy-4-estren-3-one | ChEBI |
| 17alpha-Ethynyl-17beta-hydroxy-19-norandrost-4-en-3-one | ChEBI |
| 17alpha-Ethynyl-19-nor-4-androsten-17beta-ol-3-one | ChEBI |
| 17alpha-Ethynyl-19-norandrost-4-en-17beta-ol-3-one | ChEBI |
| 17alpha-Ethynyl-19-nortestosterone | ChEBI |
| 17alpha-Ethynyl-4-estren-17-ol-3-one | ChEBI |
| 17beta-Hydroxy-19-norpregn-4-en-20-yn-3-one | ChEBI |
| 19-Nor-17-alpha-ethynyl-17-beta-hydroxy-4-androsten-3-one | ChEBI |
| 19-Nor-17-alpha-ethynylandrosten-17-beta-ol-3-one | ChEBI |
| 19-Nor-17-alpha-ethynyltestosterone | ChEBI |
| 19-Nor-17alpha-ethynyl-17beta-hydroxy-4-androsten-3-one | ChEBI |
| 19-Nor-17alpha-ethynylandrosten-17beta-ol-3-one | ChEBI |
| 19-Nor-17alpha-ethynyltestosterone | ChEBI |
| 19-Nor-ethindrone | ChEBI |
| 19-Norethisterone | ChEBI |
| 4-Estren-17alpha-ethynyl-17beta-ol-3-one | ChEBI |
| Camila | ChEBI |
| Conludag | ChEBI |
| Micronor | ChEBI |
| Micronovum | ChEBI |
| Mini-pe | ChEBI |
| Mini-pill | ChEBI |
| Norcolut | ChEBI |
| Norethisteron | ChEBI |
| Norethisteronum | ChEBI |
| Noretisterona | ChEBI |
| Noriday | ChEBI |
| Norluten | ChEBI |
| Norlutin | ChEBI |
| Primolut-N | ChEBI |
| Utovlan | ChEBI |
| Norethisterone | Kegg |
| (17a)-17-Ethynyl-17-hydroxyestra-4,8(14),9-trien-3-one | Generator |
| (17Α)-17-ethynyl-17-hydroxyestra-4,8(14),9-trien-3-one | Generator |
| 17-a-Ethynyl-17-hydroxy-4-estren-3-one | Generator |
| 17-Α-ethynyl-17-hydroxy-4-estren-3-one | Generator |
| 17-a-Ethynyl-19-norandrost-4-en-17-b-ol-3-one | Generator |
| 17-Α-ethynyl-19-norandrost-4-en-17-β-ol-3-one | Generator |
| 17-a-Ethynyl-19-nortestosterone | Generator |
| 17-Α-ethynyl-19-nortestosterone | Generator |
| 17-a-Ethynyl-4-estren-17-ol-3-one | Generator |
| 17-Α-ethynyl-4-estren-17-ol-3-one | Generator |
| 17-b-Hydroxy-19-norpregn-4-en-20-yn-3-one | Generator |
| 17-Β-hydroxy-19-norpregn-4-en-20-yn-3-one | Generator |
| 17-Hydroxy-19-nor-17-a-pregn-4-en-20-yn-3-one | Generator |
| 17-Hydroxy-19-nor-17-α-pregn-4-en-20-yn-3-one | Generator |
| 17-Hydroxy-19-nor-17a-pregn-4-en-20-yn-3-one | Generator |
| 17-Hydroxy-19-nor-17α-pregn-4-en-20-yn-3-one | Generator |
| 17a-Ethinyl-19-nortestosterone | Generator |
| 17Α-ethinyl-19-nortestosterone | Generator |
| 17a-Ethinylestra-4-en-17b-ol-3-one | Generator |
| 17Α-ethinylestra-4-en-17β-ol-3-one | Generator |
| 17a-Ethynyl-17-hydroxy-4-estren-3-one | Generator |
| 17Α-ethynyl-17-hydroxy-4-estren-3-one | Generator |
| 17a-Ethynyl-17b-hydroxy-19-norandrost-4-en-3-one | Generator |
| 17Α-ethynyl-17β-hydroxy-19-norandrost-4-en-3-one | Generator |
| 17a-Ethynyl-19-nor-4-androsten-17b-ol-3-one | Generator |
| 17Α-ethynyl-19-nor-4-androsten-17β-ol-3-one | Generator |
| 17a-Ethynyl-19-norandrost-4-en-17b-ol-3-one | Generator |
| 17Α-ethynyl-19-norandrost-4-en-17β-ol-3-one | Generator |
| 17a-Ethynyl-19-nortestosterone | Generator |
| 17Α-ethynyl-19-nortestosterone | Generator |
| 17a-Ethynyl-4-estren-17-ol-3-one | Generator |
| 17Α-ethynyl-4-estren-17-ol-3-one | Generator |
| 17b-Hydroxy-19-norpregn-4-en-20-yn-3-one | Generator |
| 17Β-hydroxy-19-norpregn-4-en-20-yn-3-one | Generator |
| 19-Nor-17-a-ethynyl-17-b-hydroxy-4-androsten-3-one | Generator |
| 19-Nor-17-α-ethynyl-17-β-hydroxy-4-androsten-3-one | Generator |
| 19-Nor-17-a-ethynylandrosten-17-b-ol-3-one | Generator |
| 19-Nor-17-α-ethynylandrosten-17-β-ol-3-one | Generator |
| 19-Nor-17-a-ethynyltestosterone | Generator |
| 19-Nor-17-α-ethynyltestosterone | Generator |
| 19-Nor-17a-ethynyl-17b-hydroxy-4-androsten-3-one | Generator |
| 19-Nor-17α-ethynyl-17β-hydroxy-4-androsten-3-one | Generator |
| 19-Nor-17a-ethynylandrosten-17b-ol-3-one | Generator |
| 19-Nor-17α-ethynylandrosten-17β-ol-3-one | Generator |
| 19-Nor-17a-ethynyltestosterone | Generator |
| 19-Nor-17α-ethynyltestosterone | Generator |
| 4-Estren-17a-ethynyl-17b-ol-3-one | Generator |
| 4-Estren-17α-ethynyl-17β-ol-3-one | Generator |
| (17-alpha)-17-Hydroxy-19-norpregn-4-en-20-yn-3-one | HMDB |
| 17-alpha-Ethynyl-17-beta-hydroxy-4-estren-3-one | HMDB |
| 17-Ethinyl-19-nor-testosterone | HMDB |
| 17-Ethynyl-17-hydroxyestr-4-en-3-one (acd/name 4.0) | HMDB |
| 17-Ethynyl-17beta-hydroxyestr-4-en-3-one | HMDB |
| 17-Ethynyl-19-nortestosterone | HMDB |
| 17-Hydroxy-(17alpha)-19-norpregn-4-en-20-yn-3-one | HMDB |
| 17-Hydroxy-17-alpha-19-norpregn-4-en-20-yn-3-one | HMDB |
| 17alpha-Ethinyl-17alpha-ethinyl-19-nortestosterone | HMDB |
| 17alpha-Ethynyl-17-hydroxy-estr-4-en-3-one | HMDB |
| 17alpha-Ethynyl-17-hydroxyest-4-en-3-one | HMDB |
| 17alpha-Ethynyl-17beta-hydroxyestr-4-en-3-one | HMDB |
| 17alpha-Ethynyl-3-oxo-4-estren-17beta-ol | HMDB |
| 17alpha-Pregn-4-en-20-yn-3-one | HMDB |
| 17beta-Hydroxy-17alpha-ethynylestr-4-en-3-one | HMDB |
| 19-Nor-17-ethinyltestosterone | HMDB |
| 19-Norethinyltestosterone | HMDB |
| Anhydrohydroxynorprogesterone | HMDB |
| Anovule | HMDB |
| Conludaf | HMDB |
| ENT | HMDB |
| Ethinyl-19-nortestosterone | HMDB |
| Ethinylnortestosterone | HMDB |
| Ethynylnortestosterone | HMDB |
| Gestest | HMDB |
| Menzol | HMDB |
| Micronett | HMDB |
| Minovlar | HMDB |
| NET | HMDB |
| Nor-q.d. | HMDB |
| Nor-QD | HMDB |
| Noralutin | HMDB |
| Noresthisterone | HMDB |
| Norethyndron | HMDB |
| Norethynodrone | HMDB |
| Norfor | HMDB |
| Norgestin | HMDB |
| Norlutate | HMDB |
| Norluton | HMDB |
| Normapause | HMDB |
| Norpregneninlone | HMDB |
| Norpregneninolone | HMDB |
| Primolut N | HMDB |
| Proluteasi | HMDB |
| Utovlar | HMDB |
| Conceplan | HMDB |
| Nor QD | HMDB |
| Norethindrone, (1 beta)-isomer | HMDB |
| Norcolute | HMDB |
| Monogest | HMDB |
| NorQD | HMDB |
| Norethindrone | ChEBI |