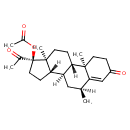
| (6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-dione | ChEBI |
| 17-Acetoxy-6alpha-methylprogesterone | ChEBI |
| 17alpha-Hydroxy-6alpha-methylprogesterone acetate | ChEBI |
| 6-alpha-Methyl-17-alpha-acetoxyprogesterone | ChEBI |
| 6-alpha-Methyl-17-alpha-hydroxyprogesterone acetate | ChEBI |
| 6alpha-Methyl-17-acetoxy progesterone | ChEBI |
| 6alpha-Methyl-17alpha-hydroxyprogesterone acetate | ChEBI |
| 6alpha-Methyl-4-pregnene-3,20-dion-17alpha-ol acetate | ChEBI |
| CBP-1011 | ChEBI |
| Depo-provera | ChEBI |
| Depot medroxyprogesterone acetate | ChEBI |
| DMPA | ChEBI |
| MAP | ChEBI |
| Medroxyacetate progesterone | ChEBI |
| Medroxyprogesterone 17-acetate | ChEBI |
| Methylacetoxyprogesterone | ChEBI |
| Metigestrona | ChEBI |
| MPA | ChEBI |
| Depo-subq provera 104 | Kegg |
| Provera | Kegg |
| (6a)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-dione | Generator |
| (6Α)-17-(acetyloxy)-6-methylpreg-4-ene-3,20-dione | Generator |
| 17-Acetoxy-6a-methylprogesterone | Generator |
| 17-Acetoxy-6α-methylprogesterone | Generator |
| 17a-Hydroxy-6a-methylprogesterone acetate | Generator |
| 17a-Hydroxy-6a-methylprogesterone acetic acid | Generator |
| 17alpha-Hydroxy-6alpha-methylprogesterone acetic acid | Generator |
| 17Α-hydroxy-6α-methylprogesterone acetate | Generator |
| 17Α-hydroxy-6α-methylprogesterone acetic acid | Generator |
| 6-a-Methyl-17-a-acetoxyprogesterone | Generator |
| 6-Α-methyl-17-α-acetoxyprogesterone | Generator |
| 6-a-Methyl-17-a-hydroxyprogesterone acetate | Generator |
| 6-a-Methyl-17-a-hydroxyprogesterone acetic acid | Generator |
| 6-alpha-Methyl-17-alpha-hydroxyprogesterone acetic acid | Generator |
| 6-Α-methyl-17-α-hydroxyprogesterone acetate | Generator |
| 6-Α-methyl-17-α-hydroxyprogesterone acetic acid | Generator |
| 6a-Methyl-17-acetoxy progesterone | Generator |
| 6Α-methyl-17-acetoxy progesterone | Generator |
| 6a-Methyl-17a-hydroxyprogesterone acetate | Generator |
| 6a-Methyl-17a-hydroxyprogesterone acetic acid | Generator |
| 6alpha-Methyl-17alpha-hydroxyprogesterone acetic acid | Generator |
| 6Α-methyl-17α-hydroxyprogesterone acetate | Generator |
| 6Α-methyl-17α-hydroxyprogesterone acetic acid | Generator |
| 6a-Methyl-4-pregnene-3,20-dion-17a-ol acetate | Generator |
| 6a-Methyl-4-pregnene-3,20-dion-17a-ol acetic acid | Generator |
| 6alpha-Methyl-4-pregnene-3,20-dion-17alpha-ol acetic acid | Generator |
| 6Α-methyl-4-pregnene-3,20-dion-17α-ol acetate | Generator |
| 6Α-methyl-4-pregnene-3,20-dion-17α-ol acetic acid | Generator |
| Depot medroxyprogesterone acetic acid | Generator |
| Medroxyacetic acid progesterone | Generator |
| Medroxyprogesterone 17-acetic acid | Generator |
| Medroxyprogesterone acetic acid | Generator |
| 6-alpha-Methyl-17alpha-hydroxyprogesterone acetate | MeSH |
| (6 alpha)-17-(Acetoxy)-6-methylpregn-4-ene-3,20-dione | MeSH |
| depo Medroxyprogesterone acetate | MeSH |
| depo-Medroxyprogesterone acetate | MeSH |
| Gestapuran | MeSH |
| 6 alpha Methyl 17alpha hydroxyprogesterone acetate | MeSH |
| Curretab | MeSH |
| Cycrin | MeSH |
| depo Provera | MeSH |
| Medroxyprogesterone 17 acetate | MeSH |
| Medroxyprogesterone 17-acetate, (6 alpha,17 alpha)-isomer | MeSH |
| Veramix | MeSH |
| DepoProvera | MeSH |
| Farlutal | MeSH |
| Medroxyprogesterone 17-acetate, (6 beta)-isomer | MeSH |
| Perlutex | MeSH |