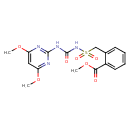
| Bensulfuron methyl | ChEBI |
| Bensulfuron methyl ester | ChEBI |
| Methyl 2-[[[[[(4,6-dimethoxy-2-pyrimidinyl)amino]carbonyl]amino]sulfonyl]methyl]benzoate | ChEBI |
| Methyl alpha-((4,6-dimethoxypyrimidin-2-yl)ureidosulfonyl)-O-toluate | ChEBI |
| Methyl alpha-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulfamoyl]-O-toluate | ChEBI |
| Methyl bensulfuron | ChEBI |
| Bensulphuron methyl | Generator |
| Bensulphuron methyl ester | Generator |
| Methyl 2-[[[[[(4,6-dimethoxy-2-pyrimidinyl)amino]carbonyl]amino]sulfonyl]methyl]benzoic acid | Generator |
| Methyl 2-[[[[[(4,6-dimethoxy-2-pyrimidinyl)amino]carbonyl]amino]sulphonyl]methyl]benzoate | Generator |
| Methyl 2-[[[[[(4,6-dimethoxy-2-pyrimidinyl)amino]carbonyl]amino]sulphonyl]methyl]benzoic acid | Generator |
| Methyl a-((4,6-dimethoxypyrimidin-2-yl)ureidosulfonyl)-O-toluate | Generator |
| Methyl a-((4,6-dimethoxypyrimidin-2-yl)ureidosulfonyl)-O-toluic acid | Generator |
| Methyl a-((4,6-dimethoxypyrimidin-2-yl)ureidosulphonyl)-O-toluate | Generator |
| Methyl a-((4,6-dimethoxypyrimidin-2-yl)ureidosulphonyl)-O-toluic acid | Generator |
| Methyl alpha-((4,6-dimethoxypyrimidin-2-yl)ureidosulfonyl)-O-toluic acid | Generator |
| Methyl alpha-((4,6-dimethoxypyrimidin-2-yl)ureidosulphonyl)-O-toluate | Generator |
| Methyl alpha-((4,6-dimethoxypyrimidin-2-yl)ureidosulphonyl)-O-toluic acid | Generator |
| Methyl α-((4,6-dimethoxypyrimidin-2-yl)ureidosulfonyl)-O-toluate | Generator |
| Methyl α-((4,6-dimethoxypyrimidin-2-yl)ureidosulfonyl)-O-toluic acid | Generator |
| Methyl α-((4,6-dimethoxypyrimidin-2-yl)ureidosulphonyl)-O-toluate | Generator |
| Methyl α-((4,6-dimethoxypyrimidin-2-yl)ureidosulphonyl)-O-toluic acid | Generator |
| Methyl a-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulfamoyl]-O-toluate | Generator |
| Methyl a-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulfamoyl]-O-toluic acid | Generator |
| Methyl a-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulphamoyl]-O-toluate | Generator |
| Methyl a-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulphamoyl]-O-toluic acid | Generator |
| Methyl alpha-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulfamoyl]-O-toluic acid | Generator |
| Methyl alpha-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulphamoyl]-O-toluate | Generator |
| Methyl alpha-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulphamoyl]-O-toluic acid | Generator |
| Methyl α-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulfamoyl]-O-toluate | Generator |
| Methyl α-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulfamoyl]-O-toluic acid | Generator |
| Methyl α-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulphamoyl]-O-toluate | Generator |
| Methyl α-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulphamoyl]-O-toluic acid | Generator |
| Methyl bensulphuron | Generator |
| Bensulphuron-methyl | Generator |
| Londax | MeSH |
| 2-((((((4,6-Dimethoxy-2-pyrimidinyl)amino)carbonyl)amino)sulfonyl)methyl)benzoic acid methyl ester | MeSH |
| Methyl bensulfuron, lithium salt | MeSH |