| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2014-08-29 05:54:33 UTC |
|---|
| Update Date | 2016-11-09 01:09:04 UTC |
|---|
| Accession Number | CHEM003185 |
|---|
| Identification |
|---|
| Common Name | Isoprene |
|---|
| Class | Small Molecule |
|---|
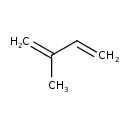
| Description | Isoprene, or 2-methyl-1,3-butadiene, is a common organic compound with the formula CH2=C(CH3)CH=CH2. It is a colorless volatile liquid. Isoprene is produced by many plants. Isoprene is incorporated into and helps stabilize cell membranes in response to heat stress, conferring some tolerance to heat spikes. Isoprene is able to cause toxic effects in the testis of rats and at multiple organ sites in mice. |
|---|
| Contaminant Sources | - Clean Air Act Chemicals
- FooDB Chemicals
- HPV EPA Chemicals
- IARC Carcinogens Group 2B
- OECD HPV Chemicals
- STOFF IDENT Compounds
- T3DB toxins
- Tobacco Smoke Compounds
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | - Cigarette Toxin
- Food Toxin
- Natural Compound
- Organic Compound
- Plant Toxin
|
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-Methyl-1,3-butadiene | ChEBI | | 2-Methylbutadiene | ChEBI | | 2-Methyldivinyl | ChEBI | | beta-Methylbivinyl | ChEBI | | CH2=C(CH3)CH=ch2 | ChEBI | | Isopentadiene | ChEBI | | Isopren | ChEBI | | Isopreno | ChEBI | | Isoterpene | ChEBI | | b-Methylbivinyl | Generator | | Β-methylbivinyl | Generator |
|
|---|
| Chemical Formula | C5H8 |
|---|
| Average Molecular Mass | 68.117 g/mol |
|---|
| Monoisotopic Mass | 68.063 g/mol |
|---|
| CAS Registry Number | 78-79-5 |
|---|
| IUPAC Name | 2-methylbuta-1,3-diene |
|---|
| Traditional Name | isoprene |
|---|
| SMILES | CC(=C)C=C |
|---|
| InChI Identifier | InChI=1S/C5H8/c1-4-5(2)3/h4H,1-2H2,3H3 |
|---|
| InChI Key | RRHGJUQNOFWUDK-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as branched unsaturated hydrocarbons. These are hydrocarbons that contains one or more unsaturated carbon atoms, and an aliphatic branch. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Hydrocarbons |
|---|
| Class | Unsaturated hydrocarbons |
|---|
| Sub Class | Branched unsaturated hydrocarbons |
|---|
| Direct Parent | Branched unsaturated hydrocarbons |
|---|
| Alternative Parents | |
|---|
| Substituents | - Branched unsaturated hydrocarbon
- Alkadiene
- Unsaturated aliphatic hydrocarbon
- Olefin
- Acyclic olefin
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | - Actin Cytoskeleton
- Cell surface
- Cytoplasm
- Cytosol
- Dendritic Process
- Endoplasmic reticulum
- Extracellular
- Extracellular matrix
- Golgi apparatus
- Lysosome
- Membrane Fraction
- Microsome
- Mitochondrial Membrane
- Mitochondrion
- Perinuclear region
- Peroxisomal Membrane
- Peroxisome
- Plasma Membrane
- Soluble Fraction
|
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | | Name | SMPDB Link | KEGG Link |
|---|
| Carbon Metabolism | Not Available | Not Available | | Metabolic Pathways | Not Available | Not Available | | Apoptosis | Not Available | map04210 | | Hmg Coa Reductase Inhibitors | Not Available | Not Available | | Nitrogen Metabolism | Not Available | Not Available | | Carotenoid Biosynthesis | Not Available | Not Available | | Retinol Metabolism | SMP00074 | map00830 | | Dna replication | Not Available | map03030 | | Terpenoid backbone biosynthesis | Not Available | map00900 | | Sphingolipid Metabolism | SMP00034 | map00600 | | Purine Metabolism | SMP00050 | map00230 | | Influenza a | Not Available | map05164 | | Fatty acid Metabolism | SMP00051 | map00071 | | Fatty Acid Biosynthesis | SMP00456 | map00061 | | Diterpenoid Biosynthesis | Not Available | Not Available | | Cell cycle | Not Available | map04110 | | Biosynthesis Of Secondary Metabolites | Not Available | Not Available | | Antifungal Agents | Not Available | Not Available | | Allograft Rejection | Not Available | Not Available |
|
|---|
| Applications | Not Available |
|---|
| Biological Roles | |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Liquid |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | -143.95°C | | Boiling Point | 34.067°C | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-9000000000-aa7bf6693faf53f2c9f8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-9000000000-79f5e8913a6e3948a925 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-9000000000-cc71ad3e01e005b392f4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-9000000000-c4e0cc5173528ae70b45 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-9000000000-748a9309c1a76a36f582 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0uxr-9000000000-b24db55f64156fae0d1d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-9000000000-13c3d4b2254b96312f2f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-9000000000-182e8f0f63d0901250e7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-9000000000-7b7c61918e48120c38a5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-9000000000-87c1aff470916cc07239 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-9000000000-87c1aff470916cc07239 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0gb9-9000000000-7033f85bf44b5b1856d1 | Spectrum | | MS | Mass Spectrum (Electron Ionization) | splash10-014i-9000000000-bcc7aa57f0fa0fd9d399 | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | 2B, possibly carcinogenic to humans. (2) |
|---|
| Uses/Sources | This is a toxic chemical found in cigarettes or generated by tobacco combustion. |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0253673 |
|---|
| FooDB ID | FDB005121 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00046784 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | CPD-9436 |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Isoprene |
|---|
| Chemspider ID | 6309 |
|---|
| ChEBI ID | 35194 |
|---|
| PubChem Compound ID | 6557 |
|---|
| Kegg Compound ID | C16521 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|