Aristolochic acid (CHEM003073)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2014-08-29 05:17:15 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2026-03-26 21:41:47 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM003073 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Aristolochic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Aristolochic acids are a family of carcinogenic, mutagenic, and nephrotoxic compounds commonly found in the Aristolochiaceae family of plants, including Aristolochia and Asarum (wild ginger), which are commonly used in Chinese herbal medicine. Aristolochic acid I is the most abundant of the aristolochic acids and is found in almost all Aristolochia species. Aristolochic acids are often accompanied by aristolactams. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
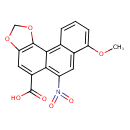
| Chemical Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | C17H11NO7 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 341.272 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 341.054 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 313-67-7 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | 6-methoxy-9-nitro-14,16-dioxatetracyclo[8.7.0.0²,⁷.0¹³,¹⁷]heptadeca-1,3,5,7,9,11,13(17)-heptaene-11-carboxylic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | birthwort | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | COC1=CC=CC2=C3C4=C(OCO4)C=C(C(O)=O)C3=C(C=C12)N(=O)=O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/C17H11NO7/c1-23-12-4-2-3-8-9(12)5-11(18(21)22)14-10(17(19)20)6-13-16(15(8)14)25-7-24-13/h2-6H,7H2,1H3,(H,19,20) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | BBFQZRXNYIEMAW-UHFFFAOYSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of organic compounds known as aristolochic acids and derivatives. These are organic heterocyclic compounds with a structure characterized by a nitrophenanthro[3,4-d][1,3]dioxole ring system substituted at position 5, 6, and 8 by a carboxyl group (or a derivative thereof), a nitro group, and a methoxy group, respectively. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Benzenoids | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Phenanthrenes and derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Aristolochic acids and derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Aristolochic acids and derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aromatic heteropolycyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | White powder. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | The carcinogenic and mutagenic effects associated with the binding of metabolites of ingested aristolochic acid (AA) to DNA have been extensively described in vitro and in vivo, resulting in the classification of AA as a genotoxic carcinogen. AA-derived DNA adducts in renal cortical and urothelial tumor tissue of patients with documented BEN, associated with the dominance of the A:T to T:A transversions in the p53 tumor suppressor gene mutational spectrum. (1) AA is a nephrotoxic and carcinogenic compound, which has been demonstrated to be genotoxic and mutagenic both in vitro and in vivo. Toxicity and carcinogenicity of the nephrotoxic compound aristolochic acid between rodents and humans suggest a species-dependent mechanism of action. AA had a comparable effect on the cell cycle in primary human and porcine cells and the rat NRK-52E cell line following 48 h exposure, also corroborated by the reduced 3H-thymidine incorporation in NRK-52E cells. In addition, DNA unwinding, suggestive of enhanced DNA damage, was observed in primary porcine cells. (2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Aristolochic acids are absorbed after oral exposure. They are metabolized to aristolactams, which are further metabolized to a cyclic N-acylnitrenium ion, a re-active intermediateforming adducts with purine bases (adenine and guanine) in DNA (dA-AAI, dG-AAI, dA-AAII, and dG-AAII). A number of cytosolic and microsomal enzymes (CYP1A1, CYP1A2, NADPH:CYP reductase, prostaglandin H synthase, DT-diaphorase, xanthine oxidase, cyclooxygenase, and NAD(P)H:quinone oxidore-ductase) are capable of bioactivating aristolochic acids to the reactive form. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | Aristolochic acid, defined as an extract of Aristolochia species comprising a mixture of aristolochic acid I and aristolochic acid II, is classified by IARC as carcinogenic to humans (Group 1). (3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Aristolochic acids are a family of carcinogenic, mutagenic, and nephrotoxic compounds commonly found in the Aristolochiaceae family of plants, including Aristolochia and Asarum (wild ginger), which are commonly used in Chinese herbal medicine. Aristolochic acid I is the most abundant of the aristolochic acids and is found in almost all Aristolochia species. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | C00001526 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Aristolochic_acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | 2149 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | 2825 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | C08469 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||