| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2013-04-25 07:56:51 UTC |
|---|
| Update Date | 2016-11-09 01:08:58 UTC |
|---|
| Accession Number | CHEM002809 |
|---|
| Identification |
|---|
| Common Name | Fluroxypyr |
|---|
| Class | Small Molecule |
|---|
| Description | Fluroxypyr is an herbicide in the class of synthetic auxins. It is used to control broadleaf weeds and woody brush. It is formulated as the 1-methylheptyl ester (fluroxypyr-MHE). |
|---|
| Contaminant Sources | - STOFF IDENT Compounds
- T3DB toxins
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | - Amine
- Ether
- Herbicide
- Organic Compound
- Organochloride
- Organofluoride
- Pesticide
- Synthetic Compound
|
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Dauko 433 | MeSH | | 4,5-Amino-3,5-dichloro-6-fluoro-2-pyridinyloxyacetic acid | MeSH | | Starane 200 | MeSH |
|
|---|
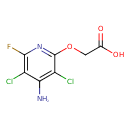
| Chemical Formula | C7H5Cl2FN2O3 |
|---|
| Average Molecular Mass | 255.031 g/mol |
|---|
| Monoisotopic Mass | 253.966 g/mol |
|---|
| CAS Registry Number | 69377-81-7 |
|---|
| IUPAC Name | 2-[(4-amino-3,5-dichloro-6-fluoropyridin-2-yl)oxy]acetic acid |
|---|
| Traditional Name | fluroxypyr |
|---|
| SMILES | NC1=C(Cl)C(OCC(O)=O)=NC(F)=C1Cl |
|---|
| InChI Identifier | InChI=1S/C7H5Cl2FN2O3/c8-3-5(11)4(9)7(12-6(3)10)15-1-2(13)14/h1H2,(H2,11,12)(H,13,14) |
|---|
| InChI Key | MEFQWPUMEMWTJP-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as polyhalopyridines. These are organic compounds containing a pyridine ring substituted at two or more positions by a halogen atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Pyridines and derivatives |
|---|
| Sub Class | Halopyridines |
|---|
| Direct Parent | Polyhalopyridines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Polyhalopyridine
- Alkyl aryl ether
- Aminopyridine
- 2-halopyridine
- Aryl chloride
- Aryl fluoride
- Aryl halide
- Heteroaromatic compound
- Amino acid or derivatives
- Amino acid
- Azacycle
- Carboxylic acid derivative
- Carboxylic acid
- Ether
- Monocarboxylic acid or derivatives
- Organic nitrogen compound
- Organohalogen compound
- Organochloride
- Organofluoride
- Organonitrogen compound
- Organooxygen compound
- Carbonyl group
- Primary amine
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Hydrocarbon derivative
- Amine
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | White powder. |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | 232°C | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0290000000-844a92785850e04fc96b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udr-0090000000-a8049ecd817cdb572874 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00di-0900000000-4f0c479ddd892433e82f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0090000000-c7dc6e99257b3a55e00d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0kai-0390000000-bb1c76365ab3655b13d2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0019-1890000000-e002e91605c408d88a6a | Spectrum | | MS | Mass Spectrum (Electron Ionization) | splash10-053r-0940000000-2c1dc60db51914c248e5 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). |
|---|
| Uses/Sources | This is a man-made compound that is used as a pesticide. |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Fluroxypyr |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 82017 |
|---|
| PubChem Compound ID | 50465 |
|---|
| Kegg Compound ID | C18858 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|