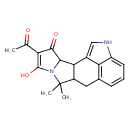
Cyclopiazonic acid (CHEM002705)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2010-05-05 19:41:24 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2026-04-03 05:06:43 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM002705 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Cyclopiazonic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Cyclopiazonic acid (CPA) is a myxotoxin originally isolated from Penicillium cyclopium and subsequently from Penicillium griseofulvum, Penicillium camembertii, Aspergillus flavus and Aspergillus versicolor. It is often found co-occurring with aflatoxins. CPA is a natural contaminant of corn, peanuts, and sunflowers, as well as various types of cheese, sausages, and salamis. It may also occur in eggs, milk and meats from animal that have consumed CPA-contaminated feed and is toxic in high concentrations. (7, 2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | C20H20N2O3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 336.384 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 336.147 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 18172-33-3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | 5-acetyl-6-hydroxy-8,8-dimethyl-7,16-diazapentacyclo[9.6.1.0²,⁹.0³,⁷.0¹⁵,¹⁸]octadeca-1(17),5,11(18),12,14-pentaen-4-one | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | 5-acetyl-6-hydroxy-8,8-dimethyl-7,16-diazapentacyclo[9.6.1.0²,⁹.0³,⁷.0¹⁵,¹⁸]octadeca-1(17),5,11(18),12,14-pentaen-4-one | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | CC(=O)C1=C(O)N2C(C3C(CC4=C5C(NC=C35)=CC=C4)C2(C)C)C1=O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/C20H20N2O3/c1-9(23)14-18(24)17-16-11-8-21-13-6-4-5-10(15(11)13)7-12(16)20(2,3)22(17)19(14)25/h4-6,8,12,16-17,21,25H,7H2,1-3H3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | RLOAZVAJNNPPDI-UHFFFAOYSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of organic compounds known as 3-alkylindoles. 3-alkylindoles are compounds containing an indole moiety that carries an alkyl chain at the 3-position. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organoheterocyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Indoles and derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Indoles | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | 3-alkylindoles | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aromatic heteropolycyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | White powder. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral, dermal, inhalation, and parenteral (contaminated drugs). (6) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Cyclopiazonic acid is potent and specific inhibitor of the endoplasmic and sarcoplasmic reticulum Ca+-dependent ATPases, which are essential for calcium reuptake in the muscle contraction-relaxation cycle. CPA blocks the calcium access channel and rigidifies a subset of transmembrane helices in a nonnative configuration that is incompatible with calcium binding. Inhibition of Ca2+-ATPases results in cell death through the activation of stress-response and apoptotic pathways within the endoplasmic reticulum and mitochondria. CPA can also induce both secretion and mRNA levels of proinflammatory cytokines, likely leading to macrophage activation and immunotoxic effects. In addition, it has been shown to be mutagenic and genotoxic. Mycotoxins are often able to enter the liver and kidney by human organic anion transporters (hOATs) and human organic cation transporters (hOCTs). They can also inhibit uptake of anions and cations by these transporters, interefering with the secretion of endogenous metabolites, drugs, and xenobiotics including themselves. This results in increased cellular accumulation of toxic compounds causing nephro- and hepatotoxicity. (1, 2, 4, 5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | LD50: 13 mg/kg (Intraperitoneal, Mouse) (3) LD50: 2.3 mg/kg (Intraperitoneal, Rat) (4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Cyclopiazonic acid (CPA) is a myxotoxin originally isolated from Penicillium cyclopium and subsequently from Penicillium griseofulvum, Penicillium camembertii, Aspergillus flavus and Aspergillus versicolor. It is often found co-occurring with aflatoxins. CPA is a natural contaminant of corn, peanuts, and sunflowers, as well as various types of cheese, sausages, and salamis. It may also occur in eggs, milk and meats from animal that have consumed CPA-contaminated feed. (7, 2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Cyclopiazonic acid affects muscle function and may also be immunotoxic, genotoxic, and mutagenic. (2, 3, 4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Animals studies have shown cyclopiazonic acid to cause symptoms such as tremors, convulsions, dyspnoea, hypokinesia, hypothermia, sedation, tachycardia, and tachypnoea. (3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Cyclopiazonic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 2908 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General References | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||