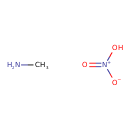
Methylammonium nitrate (CHEM002555)
| Record Information | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-08-05 17:25:25 UTC | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2016-11-09 01:08:53 UTC | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM002555 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Methylammonium nitrate | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Methylammonium nitrate is an explosive chemical formed by the mixture of methylamine and nitric acid. Nitrite is a toxic compound known to cause methemoglobinemia. (4, 6) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Structure | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | CH6N2O3 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 94.070 g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 94.038 g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 22113-87-7 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | methanamine; nitric acid | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | methylamine; nitric acid | |||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | CN.O[N+]([O-])=O | |||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/CH5N.HNO3/c1-2;2-1(3)4/h2H2,1H3;(H,2,3,4) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | PTIUDKQYXMFYAI-UHFFFAOYSA-N | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of organic compounds known as organic nitrates. These are organic compounds containing the nitrate oxoanion, with the formula NO3-. | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organic oxygen compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Organic oxoanionic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Organic nitrates | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Organic nitrates | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aliphatic acyclic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | White powder. | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (4) ; inhalation (4) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Nitrate's toxicity is a result of it's conversion to nitrite once in the body. Nitrite causes the autocatalytic oxidation of oxyhemoglobin to hydrogen peroxide and methemoglobin. This elevation of methemoglobin levels is a condition known as methemoglobinemia, and is characterized by tissue hypoxia, as methemoglobin cannot bind oxygen. (2, 5) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Intake of some amount of nitrates and nitrites is a normal part of the nitrogen cycle in humans. In vivo conversion of nitrates to nitrites can occur in the gastrointestional tract under the right conditions, significantly enhancing nitrates' toxic potency. The major metabolic pathway for nitrate is conversion to nitrite, and then to ammonia. Nitrites, nitrates, and their metabolites are excreted in the urine. (4) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | Ingested nitrate or nitrite under conditions that result in endogenous nitrosation is probably carcinogenic to humans (Group 2A). (3) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Nitrate and nitrite poisoning causes methemoglobinemia. Nitrites may cause pregnancy complications and developmental effects. They may also be carcinogenic. (4) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Nitrate and nitrite poisoning causes methemoglobinemia. Symptoms include cyanosis, cardiac dysrhythmias and circulatory failure, and progressive central nervous system (CNS) effects. CNS effects can range from mild dizziness and lethargy to coma and convulsions. (4) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | Methemoglobinemia can be treated with supplemental oxygen and methylene blue 1% solution administered intravenously slowly over five minutes followed by IV flush with normal saline. Methylene blue restores the iron in hemoglobin to its normal (reduced) oxygen-carrying state. (5) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Methylammonium nitrate | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 159924 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| General References | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||