Acetic acid (CHEM002454)
| Record Information | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-07-30 17:56:54 UTC | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2026-03-26 20:03:30 UTC | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM002454 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Acetic acid | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Acetic acid is one of the simplest carboxylic acids. It is an important chemical reagent and industrial chemical that is used in the production of plastic soft drink bottles, photographic film; and polyvinyl acetate for wood glue, as well as many synthetic fibres and fabrics. Acetic acid can be very corrosive, depending on the concentration. It is one ingredient of cigarette. In households diluted acetic acid is often used as a cleaning agent. In the food industry acetic acid is used as an acidity regulator. The acetyl group, derived from acetic acid, is fundamental to the biochemistry of virtually all forms of life. When bound to coenzyme A it is central to the metabolism of carbohydrates and fats. However, the concentration of free acetic acid in cells is kept at a low level to avoid disrupting the control of the pH of the cell contents. Acetic acid is produced and excreted by certain bacteria, notably the Acetobacter genus and Clostridium acetobutylicum. These bacteria are found universally in foodstuffs, water, and soil, and acetic acid is produced naturally as fruits and some other foods spoil. Acetic acid is also a component of the vaginal lubrication of humans and other primates, where it appears to serve as a mild antibacterial agent. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
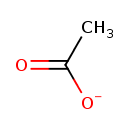
| Chemical Structure | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | C2H3O2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 59.044 g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 59.013 g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 64-19-7 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | acetate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | acetate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | CC([O-])=O | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/C2H4O2/c1-2(3)4/h1H3,(H,3,4)/p-1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | QTBSBXVTEAMEQO-UHFFFAOYSA-M | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of organic compounds known as carboxylic acids. Carboxylic acids are compounds containing a carboxylic acid group with the formula -C(=O)OH. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organic acids and derivatives | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Carboxylic acids and derivatives | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Carboxylic acids | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Carboxylic acids | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aliphatic acyclic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Endogenous | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State | Liquid | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | Colorless liquid. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (17) ; ihalation (17) ; ingestion (17) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Acetic acid is toxic due to its corrosive nature. In addition to causing skin burns and irritation to the mucous membranes, ingestion can result in severe damage to the digestive system and a potentially lethal change in the acidity of the blood. (16) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Acetic acid is is absorbed from the gastrointestinal tract and through the lungs. It is completely oxidized by the tissues, with metabolism involving the formation of ketone bodies. The products of acetic acid are used in the formation of glycogen, as intermediates of carbohydrates and fatty acid synthesis, and in cholesterol synthesis. In addition, acetic acid participates in the acetylation of amines and formation of proteins of plasma, liver, kidney, gut mucosa, muscle, and brain. (17) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | LD50: 3.53 g/kg (Oral, Rat) (20) LD50: 525 mg/kg (Intravenous, Mouse) (19) LD50: 1060 mg/kg (Dermal, Rabbit) (19) LC50: 5620 ppm over 1 hour (Inhalation, Mouse) (2) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Acetic acid gives vinegar its sour taste and pungent smell. It is an important chemical reagent and industrial chemical, used in the plastic, pharmaceutical, dye, insecticide, textile, rubber, and photographic industries. Water-free acetic acid (glacial acetic acid) is used in the production of certain fragrances. (16) Used to treat infections in the ear canal. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Concentrated acetic acid is corrosive and can cause skin burns, permanent eye damage, and irritation to the mucous membranes. Ingestion can cause severe damage to the digestive system and a potentially lethal change in the acidity of the blood. (16) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Acetic acid is corrosive and can cause skin burns and irritation to the mucous membranes. These burns or blisters may not appear until hours after exposure. (16) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | In cases of skin or eye exposure, the area should be flushed with water and burns covered with dry, sterile dressings after decontamination. If ingested, rinse mouth and administer 5 mL/kg up to 200 mL of water for dilution. Watch for signs of respiratory insufficiency and assist respiration if necessary. (1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | DBMET01513 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | HMDB0159743 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | FDB030644 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | ACET | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Acetate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | 30089 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 175 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | YMDB01010 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Earl M. Chamberlin, Warren K. Russ, Jr., George G. Hazen, “Process for preparing [1-oxo-2-cyclopentyl (or 2-isopropyl)-2-methyl-6,7-dichloro-5-indanyloxy] acetic acid.” U.S. Patent US3950408, issued April, 1928. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Link | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General References |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||