| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2009-07-21 20:28:54 UTC |
|---|
| Update Date | 2016-11-09 01:08:46 UTC |
|---|
| Accession Number | CHEM002400 |
|---|
| Identification |
|---|
| Common Name | Prazepam |
|---|
| Class | Small Molecule |
|---|
| Description | Prazepam is only found in individuals that have used or taken this drug. It is a benzodiazepine that is used in the treatment of anxiety disorders. It is a schedule IV drug in the U.S. Prazepam is believed to stimulate GABA receptors in the ascending reticular activating system. Since GABA is inhibitory, receptor stimulation increases inhibition and blocks both cortical and limbic arousal following stimulation of the brain stem reticular formation. |
|---|
| Contaminant Sources | - HMDB Contaminants - Urine
- IARC Carcinogens Group 3
- STOFF IDENT Compounds
- T3DB toxins
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | - Amide
- Amine
- Anti-Anxiety Agent
- Drug
- GABA Modulator
- Hypnotic and Sedative
- Metabolite
- Organic Compound
- Organochloride
- Synthetic Compound
|
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Centrax | Kegg | | Parke davis brand OF prazepam | HMDB | | Warner-lambert brand OF prazepam | HMDB | | Demetrin | HMDB | | Pfizer brand OF prazepam | HMDB | | Reapam | HMDB | | Lysanxia | HMDB | | mono Demetrin | HMDB | | United drug brand OF prazepam | HMDB |
|
|---|
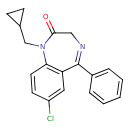
| Chemical Formula | C19H17ClN2O |
|---|
| Average Molecular Mass | 324.804 g/mol |
|---|
| Monoisotopic Mass | 324.103 g/mol |
|---|
| CAS Registry Number | 2955-38-6 |
|---|
| IUPAC Name | 7-chloro-1-(cyclopropylmethyl)-5-phenyl-2,3-dihydro-1H-1,4-benzodiazepin-2-one |
|---|
| Traditional Name | prazepam |
|---|
| SMILES | ClC1=CC2=C(C=C1)N(CC1CC1)C(=O)CN=C2C1=CC=CC=C1 |
|---|
| InChI Identifier | InChI=1S/C19H17ClN2O/c20-15-8-9-17-16(10-15)19(14-4-2-1-3-5-14)21-11-18(23)22(17)12-13-6-7-13/h1-5,8-10,13H,6-7,11-12H2 |
|---|
| InChI Key | MWQCHHACWWAQLJ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 1,4-benzodiazepines. These are organic compounds containing a benzene ring fused to a 1,4-azepine. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzodiazepines |
|---|
| Sub Class | 1,4-benzodiazepines |
|---|
| Direct Parent | 1,4-benzodiazepines |
|---|
| Alternative Parents | |
|---|
| Substituents | - 1,4-benzodiazepine
- Alpha-amino acid or derivatives
- Aryl chloride
- Aryl halide
- Monocyclic benzene moiety
- Benzenoid
- Tertiary carboxylic acid amide
- Carboxamide group
- Ketimine
- Lactam
- Carboxylic acid derivative
- Azacycle
- Propargyl-type 1,3-dipolar organic compound
- Organic 1,3-dipolar compound
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Organochloride
- Organohalogen compound
- Organic oxygen compound
- Imine
- Organic nitrogen compound
- Carbonyl group
- Organic oxide
- Organopnictogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | White powder. |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | 145-146°C | | Boiling Point | Not Available | | Solubility | 3.99e-03 g/L |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-002e-3090000000-85ecde8de7c9923c1afc | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 35V, Positive | splash10-00b9-0098000000-121cdb4df09299b2a55f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-056r-5009000000-0f619abd238690bf909e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-9022000000-7d0f4b34a0dc683c936a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-9000000000-7386d4be3880cf54c894 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0029000000-d01af5072c81e6d91456 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00xr-1097000000-243b0ed20eedd8c76c72 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9460000000-91a4eb6554df563a3898 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-0009000000-db9fa6607403d494beac | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0032-0094000000-cc4f8e8cd82551329624 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-05n3-4190000000-fa9f154eaa14bdf4214a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0009000000-33c2d52f8e6510237ee4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00yi-3095000000-d45edb96f10dbdf7642e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-001i-9070000000-47517480edb6242ad95f | Spectrum | | MS | Mass Spectrum (Electron Ionization) | splash10-00r5-3391000000-0113e86cca51d223d412 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Oral |
|---|
| Mechanism of Toxicity | Prazepam is believed to stimulate GABA receptors in the ascending reticular activating system. Since GABA is inhibitory, receptor stimulation increases inhibition and blocks both cortical and limbic arousal following stimulation of the brain stem reticular formation. |
|---|
| Metabolism | Hepatic. Prazepam is metabolised into descyclopropylmethylprazepam (also known as desmethyldiazepam) and 3-hydroxyprazepam which is further metabolised into oxazepam. [Wikipedia]
Half Life: 36-200 hours |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | 3, not classifiable as to its carcinogenicity to humans. (1) |
|---|
| Uses/Sources | For the treatment of anxiety disorders. |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | They cause slurred speech, disorientation and "drunken" behavior. They are physically and psychologically addictive. |
|---|
| Symptoms | Symptoms of overdose include somnolence, confusion, coma, and diminished reflexes. Respiration, pulse and blood pressure should be monitored. |
|---|
| Treatment | General supportive measures should be employed, along with intravenous fluids, and an adequate airway maintained. Hypotension may be combated by the use of norepinephrine or metaraminol. Dialysis is of limited value. Flumazenil (Anexate) is a competitive benzodiazepine receptor antagonist that can be used as an antidote for benzodiazepine overdose. In particular, flumazenil is very effective at reversing the CNS depression associated with benzodiazepines but is less effective at reversing respiratory depression. Its use, however, is controversial as it has numerous contraindications. It is contraindicated in patients who are on long-term benzodiazepines, those who have ingested a substance that lowers the seizure threshold, or in patients who have tachycardia or a history of seizures. As a general rule, medical observation and supportive care are the mainstay of treatment of benzodiazepine overdose. Although benzodiazepines are absorbed by activated charcoal, gastric decontamination with activated charcoal is not beneficial in pure benzodiazepine overdose as the risk of adverse effects often outweigh any potential benefit from the procedure. It is recommended only if benzodiazepines have been taken in combination with other drugs that may benefit from decontamination. Gastric lavage (stomach pumping) or whole bowel irrigation are also not recommended. |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB01588 |
|---|
| HMDB ID | HMDB0015527 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Prazepam |
|---|
| Chemspider ID | 4721 |
|---|
| ChEBI ID | 195969 |
|---|
| PubChem Compound ID | 4890 |
|---|
| Kegg Compound ID | C07366 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|