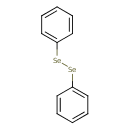| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2009-06-22 16:08:39 UTC |
|---|
| Update Date | 2016-11-09 01:08:34 UTC |
|---|
| Accession Number | CHEM001567 |
|---|
| Identification |
|---|
| Common Name | Diphenyl diselenide |
|---|
| Class | Small Molecule |
|---|
| Description | Diphenyl diselenide is a chemical compound of selenium. It is a useful reagent in organic synthesis. Selenium is a nonmetal element with the atomic number 34 and the chemical symbol Se. Selenium rarely occurs in its elemental state in nature and is usually found in sulfide ores such as pyrite, partially replacing the sulfur in the ore matrix. It may also be found in silver, copper, lead, and nickel minerals. Though selenium salts are toxic in large amounts, trace amounts of the element are necessary for cellular function in most animals, forming the active center of the enzymes glutathione peroxidase, thioredoxin reductase, and three known deiodinase enzymes. (4, 5) |
|---|
| Contaminant Sources | - IARC Carcinogens Group 3
- T3DB toxins
|
|---|
| Contaminant Type | - Aromatic Hydrocarbon
- Industrial/Workplace Toxin
- Organic Compound
- Pollutant
- Selenium Compound
- Synthetic Compound
|
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Diphenyldiselenide | MeSH |
|
|---|
| Chemical Formula | C12H10Se2 |
|---|
| Average Molecular Mass | 312.130 g/mol |
|---|
| Monoisotopic Mass | 313.911 g/mol |
|---|
| CAS Registry Number | 1666-13-3 |
|---|
| IUPAC Name | (phenyldiselanyl)benzene |
|---|
| Traditional Name | diphenyl diselenide |
|---|
| SMILES | [Se]([Se]C1=CC=CC=C1)C1=CC=CC=C1 |
|---|
| InChI Identifier | InChI=1S/C12H10Se2/c1-3-7-11(8-4-1)13-14-12-9-5-2-6-10-12/h1-10H |
|---|
| InChI Key | YWWZCHLUQSHMCL-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as benzene and substituted derivatives. These are aromatic compounds containing one monocyclic ring system consisting of benzene. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Benzene and substituted derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Monocyclic benzene moiety
- Hydrocarbon derivative
- Organoselenium compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | Orange powder. |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | 63.5°C | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0009000000-333e4185ba16a02e91ef | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0903000000-e78fc5bb8a8dabaf6d7b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0bt9-3915000000-7e7a74ec6aec2ca35aaf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0r00-0903000000-dc4ee15d46f6636b05c5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0009000000-a134aa878f4c84ad2f1b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0zfr-0900000000-a048f4303a1c6bcc39dc | Spectrum | | MS | Mass Spectrum (Electron Ionization) | splash10-0r29-8915000000-7a397647b440213a1de9 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Oral (3) ; inhalation (3) ; dermal (3) |
|---|
| Mechanism of Toxicity | Selenium readily substitutes for sulfur in biomolecules and in many biochemical reactions, especially when the concentration of selenium is high and the concentration of sulfur is low. Inactivation of the sulfhydryl enzymes necessary for oxidative reactions in cellular respiration, through effects on mitochondrial and microsomal electron transport, might contribute to acute selenium toxicity. Selenomethionine (a common organic selenium compound) also appears to randomly substitute for methionine in protein synthesis. This substitution may affect the structure and functionability of the protein, for example, by altering disulfide bridges. Inorganic forms of selenium appear to react with tissue thiols by redox catalysis, resulting in formation of reactive oxygen species and causing damage by oxidative stress. (3) |
|---|
| Metabolism | Selenium may be absorbed through inhalation and ingestion, while some selenium compounds may also be absorbed dermally. Once in the body, selenium is distributed mainly to the liver and kidney. Selenium is an essential micronutrient and is a component of glutathione peroxidase, iodothyronine 5'-deiodinases, and thioredoxin reductase. Organic selenium is first metabolized into inorganic selenium. Inorganic selenium is reduced stepwise to the intermediate hydrogen selenide, which is either incorporated into selenoproteins after being transformed to selenophosphate and selenocysteinyl tRNA or excreted into the urine after being transformed into methylated metabolites of selenide. Elemental selenium is also methylated before excretion. Selenium is primarily eliminated in the urine and feces, but certain selenium compounds may also be exhaled. (3) |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | 3, not classifiable as to its carcinogenicity to humans. (2) |
|---|
| Uses/Sources | Diphenyl diselenide is a useful reagent in organic synthesis. (5) |
|---|
| Minimum Risk Level | Chronic Oral: 0.005 mg/kg/day (1) |
|---|
| Health Effects | Chronic oral exposure to high concentrations of selenium compounds can produce a disease called selenosis. The major signs of selenosis are hair loss, nail brittleness, and neurological abnormalities (such as numbness and other odd sensations in the extremities). Animal studies have shown that selenium may also affect sperm production and the female reproductive cycle. (3) |
|---|
| Symptoms | Short-term oral exposure to high concentrations of selenium may cause nausea, vomiting, and diarrhea. Brief exposures to high levels of elemental selenium or selenium dioxide in air can result in respiratory tract irritation, bronchitis, difficulty breathing, and stomach pains. Longer-term exposure to either of these air-borne forms can cause respiratory irritation, bronchial spasms, and coughing. (3) |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Diphenyl diselenide |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 15460 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|