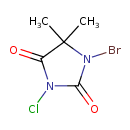
1-Bromo-3-chloro-5,5-dimethylhydantoin (CHEM001525)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-06-22 16:08:35 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2026-03-31 17:31:27 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM001525 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | 1-Bromo-3-chloro-5,5-dimethylhydantoin | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | 1-Bromo-3-chloro-5,5-dimethylhydantoin (BCDMH) is an organobromide compound. It is structurally related to hydantoin. It is a white crystalline compound with a slight bromine and acetone odor and is insoluble in water, but soluble in acetone. BCDMH is used as a solid halohydantion product for water disinfection and is an excellent source of both chlorine and bromine as it reacts slowly with water releasing hypochlorous acid and hypobromous acid along with 5,5-dimethylhydantion. It is primarily used as a chemical disinfectant for recreational water and drinking water purification. BCDMH was described as being responsible for an epidemic of irritant contact dermatitis in the UK (1983). It is prepared by first brominating, then chlorinating 5,5-dimethylhydantoin. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | C5H6BrClN2O2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 241.470 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 239.930 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 16079-88-2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | 1-bromo-3-chloro-5,5-dimethylimidazolidine-2,4-dione | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | bcdmh | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | CC1(C)N(Br)C(=O)N(Cl)C1=O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/C5H6BrClN2O2/c1-5(2)3(10)8(7)4(11)9(5)6/h1-2H3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | PIEXCQIOSMOEOU-UHFFFAOYSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of organic compounds known as hydantoins. These are heterocyclic compounds containing an imidazolidine substituted by ketone group at positions 2 and 4. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organoheterocyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Azolidines | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Imidazolidines | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Hydantoins | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aliphatic heteromonocyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | White powder. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (5) ; inhalation (5) ; dermal (5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | BCDMH likely causes tissue irritation through its breakdown products (hypochlorous acid and hypobromous acid) which, on their own, can cause local irritation. 5,5-dimethylhydantoin is also produced spontaneously from BCDMH decomposition and hydantoins are also known to be allergens. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Spontaneously reacts with water releasing hypochlorous acid and hypobromous acid along with 5,5-dimethylhydantion. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | LD50: 1390 mg/kg (Oral, Rat) (7) LD50: >2000 mg/kg (Dermal, Rabbit) (7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity (not listed by IARC). (4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Used as a solid halohydantion product for water disinfection in swimming pools. Occupational exposure to BCDMH may occur through inhalation and dermal contact with this compound at workplaces where BCDMH is produced or used. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Corrosive. Acute exposure causes irreversible eye damage and skin burns. Eye contact may cause loss of vision. Irritating to nose and throat and may be fatal if large quantities are inhaled. Harmful if absorbed through skin or swallowed. Can cause contact dermatitis through exposure in swimming pools. It is not carcinogenic or mutagenic. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Irritating to nose and throat. Can cause redness and itching of skin due to chronic skin exposure. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | EYES: irrigate opened eyes for several minutes under running water. INGESTION: do not induce vomiting. Rinse mouth with water (never give anything by mouth to an unconscious person). Seek immediate medical advice. SKIN: should be treated immediately by rinsing the affected parts in cold running water for at least 15 minutes, followed by thorough washing with soap and water. If necessary, the person should shower and change contaminated clothing and shoes, and then must seek medical attention. INHALATION: supply fresh air. If required provide artificial respiration. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 61828 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General References | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||