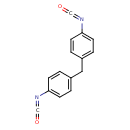
| 1,1'-Methylenebis(4-isocyanatobenzene) | ChEBI |
| 4,4'-Diisocyanatodiphenylmethane | ChEBI |
| 4,4'-Diphenylmethane diisocyanate | ChEBI |
| 4,4'-Methylene diphenyl diisocyanate | ChEBI |
| 4,4'-Methylenebis(phenyl isocyanate) | ChEBI |
| 4,4'-Methylenedi(phenyl isocyanate) | ChEBI |
| 4,4'-Methylenedi-p-phenylene diisocyanate | ChEBI |
| 4,4'-Methylenediphenyl diisocyanate | ChEBI |
| 4,4'-Methylenediphenyl isocyanate | ChEBI |
| 4,4'-Methylenediphenylene diisocyanate | ChEBI |
| 4,4'-Methylenediphenylene isocyanate | ChEBI |
| 4-4'-Diisocyanate de diphenylmethane | ChEBI |
| Bis(1,4-isocyanatophenyl)methane | ChEBI |
| Bis(4-isocyanatophenyl)methane | ChEBI |
| Bis(p-isocyanatophenyl)methane | ChEBI |
| Bis(para-isocyanatophenyl)methane | ChEBI |
| Diphenyl methane diisocyanate | ChEBI |
| Diphenylmethan-4,4'-diisocyanat | ChEBI |
| Diphenylmethane diisocyanate | ChEBI |
| Diphenylmethyl diisocyanate | ChEBI |
| MDI | ChEBI |
| MDR | ChEBI |
| Methylbisphenyl isocyanate | ChEBI |
| Methylene bisphenyl isocyanate | ChEBI |
| Methylene di-p-phenylene isocyanate | ChEBI |
| Methylene diphenyl diisocyanate | ChEBI |
| Methylenebis(4-isocyanatobenzene) | ChEBI |
| Methylenebis(4-phenyl isocyanate) | ChEBI |
| Methylenebis(4-phenylene isocyanate) | ChEBI |
| Methylenebis(p-phenyl isocyanate) | ChEBI |
| Methylenebis(p-phenylene isocyanate) | ChEBI |
| Methylenebis(para-phenyl isocyanate) | ChEBI |
| Methylenebis(para-phenylene isocyanate) | ChEBI |
| Methylenebisphenyl diisocyanate | ChEBI |
| Methylenedi-p-phenylene diisocyanate | ChEBI |
| Methylenedi-para-phenylene diisocyanate | ChEBI |
| p,P'-diphenylmethane diisocyanate | ChEBI |
| p,P'-methylenebis(phenyl isocyanate) | ChEBI |
| Para,para'-diphenylmethane diisocyanate | ChEBI |
| Para,para'-methylenebis(phenyl isocyanate) | ChEBI |
| 4,4'-Diphenylmethane diisocyanic acid | Generator |
| 4,4'-Methylene diphenyl diisocyanic acid | Generator |
| 4,4'-Methylenebis(phenyl isocyanic acid) | Generator |
| 4,4'-Methylenedi(phenyl isocyanic acid) | Generator |
| 4,4'-Methylenedi-p-phenylene diisocyanic acid | Generator |
| 4,4'-Methylenediphenyl diisocyanic acid | Generator |
| 4,4'-Methylenediphenyl isocyanic acid | Generator |
| 4,4'-Methylenediphenylene diisocyanic acid | Generator |
| 4,4'-Methylenediphenylene isocyanic acid | Generator |
| 4-4'-Diisocyanic acid de diphenylmethane | Generator |
| Diphenyl methane diisocyanic acid | Generator |
| Diphenylmethane diisocyanic acid | Generator |
| Diphenylmethyl diisocyanic acid | Generator |
| Methylbisphenyl isocyanic acid | Generator |
| Methylene bisphenyl isocyanic acid | Generator |
| Methylene di-p-phenylene isocyanic acid | Generator |
| Methylene diphenyl diisocyanic acid | Generator |
| Methylenebis(4-phenyl isocyanic acid) | Generator |
| Methylenebis(4-phenylene isocyanic acid) | Generator |
| Methylenebis(p-phenyl isocyanic acid) | Generator |
| Methylenebis(p-phenylene isocyanic acid) | Generator |
| Methylenebis(para-phenyl isocyanic acid) | Generator |
| Methylenebis(para-phenylene isocyanic acid) | Generator |
| Methylenebisphenyl diisocyanic acid | Generator |
| Methylenedi-p-phenylene diisocyanic acid | Generator |
| Methylenedi-para-phenylene diisocyanic acid | Generator |
| p,P'-diphenylmethane diisocyanic acid | Generator |
| p,P'-methylenebis(phenyl isocyanic acid) | Generator |
| Para,para'-diphenylmethane diisocyanic acid | Generator |
| Para,para'-methylenebis(phenyl isocyanic acid) | Generator |
| Diphenylmethane-4,4'-diisocyanic acid | Generator |
| 1,1-Methylenebis(phenyl)diisocyanate | MeSH |
| 4,4'-Methylenebis(phenylisocyanate) | MeSH |
| 4,4'-Methylene bisphenyl diisocyanate | MeSH |
| Diphenylmethane-4,4'-diisocyanate | MeSH |
| Methylenebis(phenyl isocyanate) | MeSH |