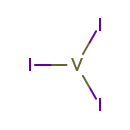
Vanadium(III) iodide (CHEM001395)
| Record Information | ||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | |||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-06-19 21:59:04 UTC | |||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2016-11-09 01:08:32 UTC | |||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM001395 | |||||||||||||||||||||||||||||||||||||||||||||
| Identification | ||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Vanadium(III) iodide | |||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | |||||||||||||||||||||||||||||||||||||||||||||
| Description | Vanadium(III) iodide is an iodide of vanadium. Vanadium is a transition metal with the chemical symbol V and atomic number 23. The element usually combines with other elements such as oxygen, sodium, sulfur, or chloride, and occurs naturally in about 65 different minerals and in fossil fuel deposits. Vanadium is found in many organisms, and is used by some life forms as an active center of enzymes. (7, 8) | |||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| |||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Structure | ||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | I3V | |||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 431.655 g/mol | |||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 431.657 g/mol | |||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 15513-94-7 | |||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | triiodovanadium | |||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | triiodovanadium | |||||||||||||||||||||||||||||||||||||||||||||
| SMILES | I[V](I)I | |||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/3HI.V/h3*1H;/q;;;+3/p-3 | |||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | AAWFOGYSSVYINI-UHFFFAOYSA-K | |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | ||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of inorganic compounds known as transition metal iodides. These are inorganic compounds in which the largest halogen atom is Iodine, and the heaviest metal atom is a transition metal. | |||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Inorganic compounds | |||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Mixed metal/non-metal compounds | |||||||||||||||||||||||||||||||||||||||||||||
| Class | Transition metal salts | |||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Transition metal iodides | |||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Transition metal iodides | |||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | ||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| |||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | ||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | |||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | |||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| |||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | ||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | |||||||||||||||||||||||||||||||||||||||||||||
| Appearance | Black solid. | |||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| |||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| |||||||||||||||||||||||||||||||||||||||||||||
| Spectra | ||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| |||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | ||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (7) ; inhalation (7) ; dermal (7) | |||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Vanadium damages alveolar macrophages by decreasing the macrophage membrane integrity, thus impairing the cells' phagocytotic ability and viability. The pentavalent form of vanadium, vanadate, is a potent inhibitor of the Ca+-ATPase and Na+,K+-ATPase of plasma membranes, which decreases intracellular ATP concentration. Vanadium is also believed to induce the production of reactive oxygen species. This may damage DNA and also cause oxidative stress, which can damage the reproductive system. Vanadium also inhibits protein tyrosine phosphatases, producing insulin-like effects. (7, 1, 2, 3, 4, 5) | |||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Vanadium is absorbed mainly via inhalation, though small amounts can be absorbed through the skin and gastrointestional tract. It is rapidly distributed in the plasma, mainly to the kidney, liver, lungs, heart, bone, where it tends to accumulate. With the help of cytochrome P-450 enzymes, it can interconvert between its two oxidation states, vanadyl (V+4) and vanadate (V+5). Both states of vanadium can reversibly bind to transferrin protein in the blood and then be taken up into erythrocytes. Vanadium is excreted mainly in the urine. (7) | |||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). | |||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Acute Inhalation: 0.0002 mg/m3 (6) Intermediate Oral: 0.003 mg/kg/day (6) | |||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Breathing high levels of vanadium affects the lungs, throat, and eyes. Ingestion of vanadium may cause kidney and liver damage, birth defects, or death. (7) | |||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Inhalation of vanadium causes lung irritation, coughing, wheezing, chest pain, runny nose, and a sore throat. (7) | |||||||||||||||||||||||||||||||||||||||||||||
| Treatment | EYES: irrigate opened eyes for several minutes under running water. INGESTION: do not induce vomiting. Rinse mouth with water (never give anything by mouth to an unconscious person). Seek immediate medical advice. SKIN: should be treated immediately by rinsing the affected parts in cold running water for at least 15 minutes, followed by thorough washing with soap and water. If necessary, the person should shower and change contaminated clothing and shoes, and then must seek medical attention. INHALATION: supply fresh air. If required provide artificial respiration. | |||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | ||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | ||||||||||||||||||||||||||||||||||||||||||||||
| External Links | ||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Vanadium(III) iodide | |||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 3627252 | |||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| General References | Not Available | |||||||||||||||||||||||||||||||||||||||||||||