Aluminum butylate (CHEM001337)
| Record Information | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-06-19 21:58:58 UTC | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2026-04-05 19:52:01 UTC | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM001337 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Aluminum butylate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Aluminum butylate is a chemical compound of aluminum. Aluminum is the most abundant metal in the earth's crust and is always found combined with other elements such as oxygen, silicon, and fluorine. (6, 7) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
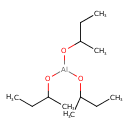
| Chemical Structure | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | C12H27AlO3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 246.323 g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 246.178 g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 2269-22-9 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | tris(butan-2-yloxy)alumane | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | tris(sec-butoxy)alumane | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | CCC(C)O[Al](OC(C)CC)OC(C)CC | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/3C4H9O.Al/c3*1-3-4(2)5;/h3*4H,3H2,1-2H3;/q3*-1;+3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | WOZZOSDBXABUFO-UHFFFAOYSA-N | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of organic compounds known as organic oxides. These are organic compounds containing an oxide group. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organic oxygen compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Organic oxides | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Organic oxides | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | White powder. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (6) ; inhalation (6) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | The main target organs of aluminum are the central nervous system and bone. Aluminum binds with dietary phosphorus and impairs gastrointestinal absorption of phosphorus. The decreased phosphate body burden results in osteomalacia (softening of the bones due to defective bone mineralization) and rickets. Aluminum's neurotoxicity is believed to involve several mechanisms. Changes in cytoskeletal protein functions as a results of altered phosphorylation, proteolysis, transport, and synthesis are believed to be one cause. Aluminum may induce neurobehavioral effects by affecting permeability of the blood-brain barrier, cholinergic activity, signal transduction pathways, lipid peroxidation, and impair neuronal glutamate nitric oxide-cyclic GMP pathway, as well as interfere with metabolism of essential trace elements because of similar coordination chemistries and consequent competitive interactions. It has been suggested that aluminum's interaction with estrogen receptors increases the expression of estrogen-related genes and thereby contributes to the progression of breast cancer (1), but studies have not been able to establish a clear link between aluminum and increased risk of breast cancer (3). Certain aluminum salts induce immune responses by activating inflammasomes. (6, 1, 2) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Aluminum is poorly absorbed following either oral or inhalation exposure and is essentially not absorbed dermally. The bioavailability of aluminum is strongly influenced by the aluminum compound and the presence of dietary constituents which can complex with aluminum and enhance or inhibit its absorption. Aluminum binds to various ligands in the blood and distributes to every organ, with highest concentrations found in bone and lung tissues. In living organisms, aluminum is believed to exist in four different forms: as free ions, as low-molecular-weight complexes, as physically bound macromolecular complexes, and as covalently bound macromolecular complexes. Absorbed aluminum is excreted principally in the urine and, to a lesser extent, in the bile, while unabsorbed aluminum is excreted in the faeces. (6) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | Not listed by IARC. IARC classified aluminum production as carcinogenic to humans (Group 1), but did not implicate aluminum itself as a human carcinogen. (5) A link between use of aluminum-containing antiperspirants and increased risk of breast cancer has been proposed (1), but studies have not been able to establish a clear link (3). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Intermediate Oral: 1.0 mg/kg/day (4) Chronic Oral: 1.0 mg/kg/day (4) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Aluminum targets the nervous system and causes decreased nervous system performance and is associated with altered function of the blood-brain barrier. The accumulation of aluminum in the body may cause bone or brain diseases. High levels of aluminum have been linked to Alzheimer's disease. A small percentage of people are allergic to aluminium and experience contact dermatitis, digestive disorders, vomiting or other symptoms upon contact or ingestion of products containing aluminium. (6, 7) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Inhalating aluminum dust causes coughing and abnormal chest X-rays. A small percentage of people are allergic to aluminium and experience contact dermatitis, digestive disorders, vomiting or other symptoms upon contact or ingestion of products containing aluminium. (6, 7) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 61289 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General References | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||