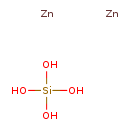
Zinc silicate (CHEM001249)
| Record Information | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-06-19 21:58:49 UTC | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2016-11-09 01:08:29 UTC | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM001249 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Zinc silicate | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Zinc silicate is a chemical compound of zinc. Zinc is a metallic element with the atomic number 30. It is found in nature most often as the mineral sphalerite. Though excess zinc in harmful, in smaller amounts it is an essential element for life, as it is a cofactor for over 300 enzymes and is found in just as many transcription factors. (2, 3) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Structure | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | H4O4SiZn2 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 226.933 g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 223.846 g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 13597-65-4 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | silicic acid dizinc | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | silicic acid dizinc | |||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | [Zn].[Zn].O[Si](O)(O)O | |||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/H4O4Si.2Zn/c1-5(2,3)4;;/h1-4H;; | |||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | COWOWCJDIGPUDT-UHFFFAOYSA-N | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of inorganic compounds known as transition metal silicates. These are inorganic compounds in which the largest oxoanion is silicate, and in which the heaviest atom not in an oxoanion is a transition metal. | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Inorganic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Mixed metal/non-metal compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Transition metal oxoanionic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Transition metal silicates | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Transition metal silicates | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | White powder. | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (3) ; inhalation (3) ; dermal (3) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Anaemia results from the excessive absorption of zinc suppressing copper and iron absorption, most likely through competitive binding of intestinal mucosal cells. Unbalanced levels of copper and zinc binding to Cu,Zn-superoxide dismutase has been linked to amyotrophic lateral sclerosis (ALS). Stomach acid dissolves metallic zinc to give corrosive zinc chloride, which can cause damage to the stomach lining. Metal fume fever is thought to be an immune response to inhaled zinc. (2, 3, 1) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Zinc can enter the body through the lungs, skin, and gastrointestinal tract. Intestinal absorption of zinc is controlled by zinc carrier protein CRIP. Zinc also binds to metallothioneins, which help prevent absorption of excess zinc. Zinc is widely distributed and found in all tissues and tissues fluids, concentrating in the liver, gastrointestinal tract, kidney, skin, lung, brain, heart, and pancreas. In the bloodstream zinc is found bound to carbonic anhydrase in erythrocytes, as well as bound to albumin, _2-macroglobulin, and amino acids in the the plasma. Albumin and amino acid bound zinc can diffuse across tissue membranes. Zinc is excreted in the urine and faeces. (3) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Intermediate Oral: 0.3 mg/kg/day (5) Chronic Oral: 0.3 mg/kg/day (5) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Chronic exposure to zinc causes anemia, atazia, lethargy, and decreases the level of good cholesterol in the body. It is also believed to cause pancreatic and reproductive damage. (3) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Ingestion of large doses of zinc causes stomach cramps, nausea, and vomiting. Acute inhalation of large amounts of zinc causes metal fume fever, which is characterized by chills, fever, headache, weakness, dryness of the nose and throat, chest pain, and coughing. Dermal contact with zinc results in skin irritation. (3) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | Zinc poisoning is treated symptomatically, often by administering fluids such as water or milk, or with gastric lavage. (3) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Zinc silicate | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 159687 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| General References | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||