Chromium(III) acetate (CHEM001243)
| Record Information | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-06-19 21:58:48 UTC | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2026-04-05 17:48:27 UTC | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM001243 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Chromium(III) acetate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Chromium(III) acetate is a chemical compound of chromium. Chromium is a chemical element which has the symbol Cr and atomic number 24. It is found naturally occuring in rocks, animals, plants, and soil, and is usually mined as chromite ore. Chromium is most toxic in its +6 oxidation state (chromium(VI)) due to its greater ability to enter cells and higher redox potential. Trivalent chromium (chromium(III)) however, is biologically necessary for sugar and lipid metabolism in humans. (6) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
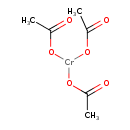
| Chemical Structure | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | C6H9CrO6 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 229.128 g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 228.980 g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 1066-30-4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | bis(acetyloxy)chromio acetate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | chromic acetate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | CC(=O)O[Cr](OC(C)=O)OC(C)=O | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/3C2H4O2.Cr/c3*1-2(3)4;/h3*1H3,(H,3,4);/q;;;+3/p-3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | WYYQVWLEPYFFLP-UHFFFAOYSA-K | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of organic compounds known as carboxylic acids. Carboxylic acids are compounds containing a carboxylic acid group with the formula -C(=O)OH. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organic acids and derivatives | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Carboxylic acids and derivatives | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Carboxylic acids | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Carboxylic acids | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | White powder. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (5) ; inhalation (5) ; dermal (5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Trivalent chromium may also form complexes with peptides, proteins, and DNA, resulting in DNA-protein crosslinks, DNA strand breaks, DNA-DNA interstrand crosslinks, chromium-DNA adducts, chromosomal aberrations and alterations in cellular signaling pathways. It has been shown to induce carcinogenesis by overstimulating cellular regulatory pathways and increasing peroxide levels by activating certain mitogen-activated protein kinases. It can also cause transcriptional repression by cross-linking histone deacetylase 1-DNA methyltransferase 1 complexes to CYP1A1 promoter chromatin, inhibiting histone modification. Chromium may increase its own toxicity by modifying metal regulatory transcription factor 1, causing the inhibition of zinc-induced metallothionein transcription. (1, 5, 2, 3, 4) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Chromium is absorbed from oral, inhalation, or dermal exposure and distributes to nearly all tissues, with the highest concentrations found in kidney and liver. Bone is also a major storage site and may contribute to long-term retention. Hexavalent chromium's similarity to sulfate and chromate allow it to be transported into cells via sulfate transport mechanisms. Inside the cell, hexavalent chromium is reduced first to pentavalent chromium, then to trivalent chromium by many substances including ascorbate, glutathione, and nicotinamide adenine dinucleotide. Chromium is almost entirely excreted with the urine. (1, 5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | LD50: 2365 mg/kg/day (Oral, Rat) (5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | 3, not classifiable as to its carcinogenicity to humans. (7) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Chromium in its trivalent state is not very toxic. It may be oxidized to hexavalent chromium, a known carcinogen. Hexavalent chromium has also been shown to affect reproduction and development. (1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Chromium in its trivalent state is not very toxic, but it may be oxidized to hexavalent chromium. Breathing hexavalent chromium can cause irritation to the lining of the nose, nose ulcers, runny nose, and breathing problems, such as asthma, cough, shortness of breath, or wheezing. Ingestion of hexavalent chromium causes irritation and ulcers in the stomach and small intestine, as well as anemia. Skin contact can cause skin ulcers. (5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | There is no know antidote for chromium poisoning. Exposure is usually handled with symptomatic treatment. (5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Chromium(III) acetate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 14012 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General References | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||