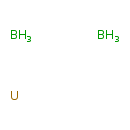Uranium boride (CHEM001002)
| Record Information | ||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | |||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-06-19 21:58:23 UTC | |||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2016-11-09 01:08:25 UTC | |||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM001002 | |||||||||||||||||||||||||||||||||||||||||||||
| Identification | ||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Uranium boride | |||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | |||||||||||||||||||||||||||||||||||||||||||||
| Description | Uranium boride is a boride of uranium. Uranium is a chemical element that has the symbol U and atomic number 92. It is a normal part of rocks, soil, air, and water, and occurs in nature in the form of minerals. (5, 6) | |||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| |||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Structure | ||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | B2H6U | |||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 265.700 g/mol | |||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 266.116 g/mol | |||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 12007-36-2 | |||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | diborane uranium | |||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | diborane uranium | |||||||||||||||||||||||||||||||||||||||||||||
| SMILES | B.B.[U] | |||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/2BH3.U/h2*1H3; | |||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | WCAPKLNAHLFYQI-UHFFFAOYSA-N | |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | ||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of inorganic compounds known as miscellaneous mixed metal/non-metals. These are inorganic compounds containing non-metal as well as metal atoms but not belonging to afore mentioned classes. | |||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Inorganic compounds | |||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Mixed metal/non-metal compounds | |||||||||||||||||||||||||||||||||||||||||||||
| Class | Miscellaneous mixed metal/non-metals | |||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Miscellaneous mixed metal/non-metals | |||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | ||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| |||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | ||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | |||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | |||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| |||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | ||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | |||||||||||||||||||||||||||||||||||||||||||||
| Appearance | White powder. | |||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| |||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| |||||||||||||||||||||||||||||||||||||||||||||
| Spectra | ||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| |||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | ||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (6) ; inhalation (6) ; dermal (6) | |||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Uranium is combined with either bicarbonate or a plasma protein in the blood but once in the kidney, it is released and forms complexes with phosphate ligands and proteins in the tubular wall, causing damage. Uranium may also inhibit both sodium transport-dependent and independent ATP utilization and mitochondrial oxidative phosphorylation in the renal proximal tubule. Uranium causes respiratory diseases by damaging alveolar epithelium type II cells in the lungs. Uranium induces c-Jun N-terminal kinase (JNK) and p38 mitogen-activated protein kinase (p38 MAPK) activation, which in turn induces tumor necrosis factor alpha (TNF-alpha) secretion and generates and inflammatory response in the lungs. Studies have shown that the more soluble the uranium salt, the more toxic it is. Ionizing radiation produced by uranium damages the DNA, resulting in gene mutations and chromosomal aberrations. This can both both initiate and promote carcinogenesis, and interfere with reproduction and development. (6, 1) | |||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Uranium is absorbed in low amounts via oral, inhalation, and dermal routes. Uranium in body fluids generally exists as the uranyl ion (UO2)2+ complexed with anions, such as citrate and bicarbonate, or plasma proteins. Uranium preferentially distributes to bone, liver, and kidney. The large majority of uranium that enters the body is not absorbed and is eliminated from the body via the urine and faeces. (5) | |||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | Uranium: Group 1, carcinogenic to humans (4) | |||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Intermediate Inhalation: 0.0004 mg/m3 (Soluble uranium salts) (3) Chronic Inhalation: 0.0003 mg/m3 (Soluble uranium salts) (3) Intermediate Oral: 0.002 mg/kg/day (Soluble uranium salts) (3) Intermediate Inhalation: 0.008 mg/m3 (Insoluble uranium compounds) (3) | |||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Uranium primarily damages the kidney, but may also damage the lungs, central nervous system, and immune system. Uranium's radioactivity is believed to damage the DNA, resulting in carcinogenic effects and reproductive and developmental damage. (5, 6) | |||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Ingestion of uranium may cause vomiting and diarrhea. (5) | |||||||||||||||||||||||||||||||||||||||||||||
| Treatment | EYES: irrigate opened eyes for several minutes under running water. INGESTION: do not induce vomiting. Rinse mouth with water (never give anything by mouth to an unconscious person). Seek immediate medical advice. SKIN: should be treated immediately by rinsing the affected parts in cold running water for at least 15 minutes, followed by thorough washing with soap and water. If necessary, the person should shower and change contaminated clothing and shoes, and then must seek medical attention. INHALATION: supply fresh air. If required provide artificial respiration. | |||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | ||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | ||||||||||||||||||||||||||||||||||||||||||||||
| External Links | ||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| General References | Not Available | |||||||||||||||||||||||||||||||||||||||||||||